Keywords
Abstract
The review addresses simple and efficient approaches to the rational synthesis of conjugates of oligonucleotides with functional molecules (FMs), including active low-molecular-weight compounds and macromolecules of various natures. These compounds can possess high reactivity, catalytic activity, or affinity for nucleic acid (NA) targets (e.g., intercalators, ligands exhibiting NA affinity); they can provide visualization (reporter groups, fluorophores), intracellular transport, desired cellular distribution, and related functions. The principles of "oligonucleotide construction set" are formulated as an algorithm taking account of specific features of conjugate formation depending on the structure of both the nucleic acid component and the FM component in terms of the pre-synthetic (direct) and postsynthetic (indirect) strategies. Unlike the existing reviews, which are most often devoted to one or a few conjugation techniques or conjugates of a particular type, the present review demonstrates the whole diversity of interactions between oligonucleotides and various compounds. The review covers both classic and modern methods that employ a minimal optimal set of efficient reactions and reagents that make it possible to preserve the functional properties of both components (NA and FM) and achieve the desired action of the conjugate on biological targets.
The bibliography includes 420 references.
1. Introduction
Oligonucleotides, that is, nucleic acid (NA) fragments, are currently used in various fields of biotechnology, molecular biology, and medicine. These compounds are indispensable for the diagnosis of various diseases and have a huge therapeutic potential, first of all, as the base for gene-targeted drugs as well as antiviral, anticancer, and immunomodulating agents. All over the world, more than twenty therapeutic agents in which active ingredients are oligonucleotides or their conjugates with biologically active functional molecules have already been approved for use.[1-6] *
Currently, the synthesized oligonucleotides (ribonucleic and deoxyribonucleic acid fragments and their phosphate or 2'-modified analogues, etc.) meet the demands of numerous research and practical applications. Their synthesis if often a routine automated procedure, although requiring skilled personnel and high-quality set of reagents. This became possible owing to studies of several research groups.[7] For example, in the 1950s, A.M.Michelson and A.R.Todd[8] laid the basis for the H-phosphonate and phosphotriester methods for oligonucleotide synthesis; P.T.Gilham and H.G.Khorana[9] developed the phosphodiester method; and in the next decade, a research group headed by R.L.Letsinger[10][11] and C.B.Reese[12] described the phosphotriester method. In the 1970s, R.L.Letsinger et al.[13] proposed a phosphite triester method for the formation of internucleoside links in which they used much more reactive nucleoside derivatives based on trivalent phosphorus. Among the specialists who actively facilitated the development and shaping up of oligonucleotide synthesis,[14-22] mention should be made of K.K.Ogilvie and S.L.Beaucage and Russian scientists: Z.A.Shabarova, O.G.Chakhmakhcheva, M.N.Kolosov, V.F.Zarytova, and D.G.Knorre. The scope of research interests of these scientists is quite extensive, ranging from protecting groups and the search for activating reagents to the development of new methods and improvement of synthetic processes. Relying on early works, research team headed by M.H.Caruthers[23][24] reported a solid-phase synthesis of oligonucleotides. The final choice of 2-cyanoethyl as a protecting group at the phosphorus atom[25] resulted in the type of nucleotide phosphoramidites that is commonly accepted now.
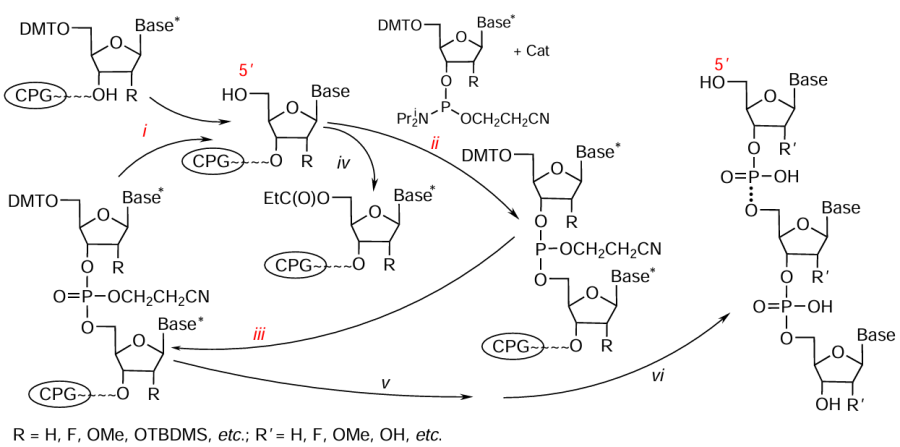
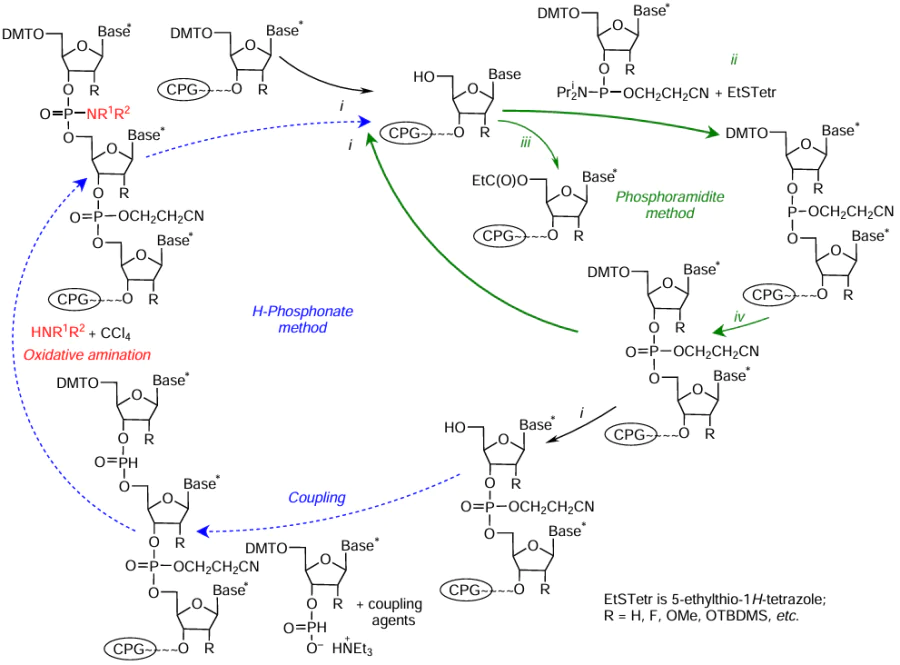
The automated oligonucleotide synthesis by the phosphoramidite method[26-30] produces both native and modified NA moieties. The synthetic cycle for addition of a single nucleotide unit includes three key steps (i – iii in Fig. 1), detritylation, coupling, and oxidation, which provide elongation of the oligonucleotide chain. The auxiliary capping step (iv) is necessary to block shortened oligomers with n – 1, n – 2, etc., formed as by-products due to incomplete coupling in the synthesis of the backbone with the target length n. The NA synthesis is performed on solid supports such as controlled pore glass or macroporous polystyrene, which allows deliberate change in the reactional or functional medium around the support in each step, thus ensuring the formation of the target oligonucleotide or oligonucleotide conjugate with a specified sequence and desired functions.
Thus, the solid-phase phosphoramidite method for the synthesis of oligonucleotides (both native ones and modified at the heterocyclic bases or sugar – phosphate backbone) and their conjugates with various functional molecules became available and widely used in the 1980s. Oligonucleotides conjugated with FMs promoted effective solution of both research (physicochemical, molecular-biological, biochemical) and practice-oriented diagnostic and therapeutic problems in medicine.
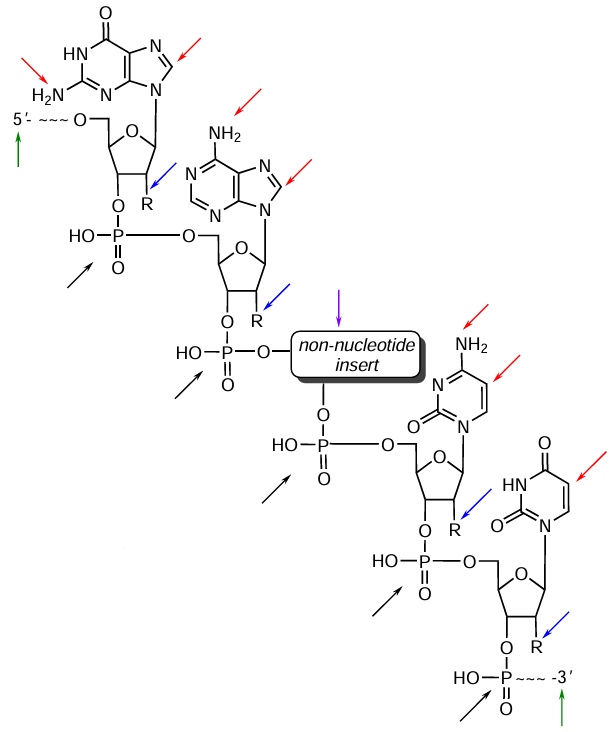
Depending on the task set, an important issue is the position of FM in the oligonucleotide chain. As a rule, FMs are introduced at the following sites: the heterocyclic base, 2'-position of ribose, internucleoside phosphate linkages, the internal part of the oligonucleotide chain (as non-nucleotide inserts), and 5'- or 3'-terminal moieties of NA (Fig. 2). For the introduction of FMs into oligonucleotides, various modifiers are used as building blocks. They represent phosphoramidites and/or solid supports of either nucleotide or non-nucleotide nature containing various groups and provide targeted functionalization of NA chain at specified positions. It is noteworthy that, despite the presence of commercial products, precursors bearing functional groups can be synthesized, if necessary, using procedures described in the literature, besides the use of several modifiers in the synthesis makes it possible to obtain not only mono- but also multi-modified oligonucleotides.
Generally, approaches to the synthesis of conjugates are distinguished are classified into pre-synthetic (I) and post-synthetic (II) ones (Fig. 3). The former comprises direct introduction of FMs during the automated synthesis, while the latter is based on indirect addition of FMs, i.e., introduction of a reactive group into the oligonucleotide during automated synthesis followed by post-synthetic functionalization of this group. Description, benefits, and details of both approaches are summarized in Table 1.
Among the first examples of application of oligonucleotide – FM conjugates, there were works on complementary-addressed modification, a fundamental versatile method for targeted action on DNA or RNA, the idea of which was first proposed back in 1967 by N.I.Grineva and co-workers.[31] It was shown that oligonucleotides bearing chemically reactive groups are capable of complementary interactions with the NA target, which ensures the targeted modification.[32-40] Studies devoted to the synthesis of oligonucleotides containing fluorescein dye were actively published in the late 1980s and the early 1990s (e.g.[41-45]). Apart from fluorescein, a popular molecule that started to be used to produce oligonucleotide conjugates by automated synthesis is hydrophobic cholesterol.[46-48] Even at that time, it was clear that enhancement of the efficiency of oligonucleotide penetration into cells and correction of the cellular distribution are crucial factors for the development of oligonucleotide-based therapeutic agents. The elaboration of procedures and approaches to the introduction of various biologically active compounds [transport molecules (cholesterol, folic acid, carbohydrates, α-tocopherol, cell-penetrating peptides, etc.), fluorescent and spin labels, peptide or peptide-like fragments with various biological activities, etc.] into oligonucleotides (aptamers, small interfering RNAs, microRNAs, gene-targeted and antisense oligonucleotides, diagnostic probes, and other biologically significant nucleic acids) provided the possibility of considerable variation of their properties. It became possible to determine the specific features of the interaction of these conjugated with NA targets or proteins, visualize their location in cells and/or organs, increase their cellular uptake, or correct the pharmacokinetic properties upon the distribution in the body (e.g.,[49-68][69-74]). The progress in the oligonucleotide chemistry markedly expanded the scope of applications for NA-based agents for both basic research and practical purposes. The first comprehensive reviews devoted to the synthesis of modified oligonucleotides and their conjugates[75-79] appeared back in the 1990s. However, there is still no commonly accepted and clear strategy for rational planning of synthesis of the desired oligonucleotide derivative that would meet the functional objectives of the experiment or provide the expected biological outcome.
This review analyses diverse conjugation reactions of oligonucleotides with various FMs and formulates the principles behind the ‘oligonucleotide construction set’ concept. This concept serves as a versatile strategy for the design of an optimal synthetic route toward oligonucleotide conjugates with integrated target FM residues using classic phosphoramidite method. Rational approaches to the synthesis imply the following stages:
(1) analysis of the FM structure in order to identify the most preferable derivative (containing, for example, amino, azido, hydroxyl, alkynyl, or another group) that can be prepared (if necessary) with high selectivity, with the involved reactions being efficient, the required effort and resource expenditure being low, and the biological function of the initial molecule being preserved;
(2) selection of suitable options for modification of the oligonucleotide component taking account of its activity toward the specific interaction with biotargets and considering the FM derivative determined in the first stage;
(3) selection of the optimal conjugation method (reagents and reaction conditions) to ensure the stability, functionality, and biocompatibility of the final product.
The analysis of existing approaches performed in this review is focused on the methods that give target conjugates using the minimal optimal set of knowingly effective commercially available reagents and processes involving them. Additionally, we consider convenient and effective protocols for the modification of oligonucleotides with unique functional molecules using lab-made modifiers that are not commercially manufactured, but can be prepared at dedicated laboratories.** The analyzed approaches demonstrate the ways to mitigate the potential synthetic obstacles faced in the design of target oligonucleotide conjugates with FM possessing specified properties.
* In this review, the term ‘conjugate’ implies a molecular construct consisting of a nucleic acid (or oligonucleotide) moiety and a functional molecule (FM) (chemical compound with definite properties that allow the molecule to perform a specific role in chemical, biological, and physical properties), where the acronym FM may refer to either a whole molecule or a part of the molecule bound to oligonucleotide by a covalent bond either directly or through a linker group.
** Lab-made modifiers are single chemical compounds designed to meet unique challenges related to the production of desired conjugates.
2. Concepts of the synthesis of conjugates of oligonucleotides with functional molecules
As noted above, conjugates possessing various functions are obtained using pre- and post-synthetic approaches (see Table 1). Irrespective of the chosen strategy for the automated synthesis of the target oligonucleotide sequence, it is necessary to use modifiers that either ensure the direct FM attachment to the desired position of the oligomer (pre-synthetic approach, see Section 3) or contain a reaction centre that is inert during the synthesis, but is capable of further functionalization for the targeted introduction of FM moiety (post-synthetic approach, see Section 4). The post-synthesis approach implies introduction of reactive groups into both the oligonucleotide and FM, in such a way that these groups selectively react with each other under specific conditions (click-reaction, thiol – maleimide coupling, Sonogashira reaction, etc.). These approaches are applicable only in the case where structural, physicochemical, and molecular-biological characteristics of FMs do not change upon modification. The broad selection of available reactive groups and introduction methods makes it possible to choose the optimal option for the synthesis of derivatives that retain the functional properties of the starting molecule. It is also important to note that the post-synthetic approach can be implemented in two ways, in particular, as synthesis on a support or in solution. The choice in favour of either of these options depends on the FM stability during the final deprotection of the oligonucleotide (derivative or conjugate) and type of post-synthetic treatment.
Chemical companies such as Lumiprobe, Primetech, Glen Research, Hongene, ChemGenes, Merck, and so on (see the company websites[80-85]) supply nucleotide and non-nucleotide modifiers for various strategies, which considerably facilitates the synthesis of the desired conjugates. However, only the most popular reagents and FMs that have proved to be effective for a wide range of fundamental and applied studies are currently available in the market. Meanwhile, quite a few research groups are continuously engaged in the development of new procedures for the laboratory synthesis of various NA modifiers.
3. Pre-synthetic approach: conjugate formation during oligonucleotide synthesis
The pre-synthetic approach (see Table 1) can be described as a facile method for the introduction of various functionally significant molecules into oligonucleotides, since this method implies that automated synthesis directly uses ready (i.e., previously obtained) commercial or lab-made FM-containing modifiers.
The modifying reagents for the synthesis of oligonucleotide conjugates can be divided into three groups:
(1) terminal phosphoramidite reagents that provide FM attachment to the 5'-end of the oligonucleotide, thus precluding the possibility of further chain elongation;
(2) internal (linear and branched) phosphoramidites: modifiers that are introduced into any chosen position of oligonucleotide;
(3) solid supports that allow for modification of the 3'-end of the oligonucleotide chain.
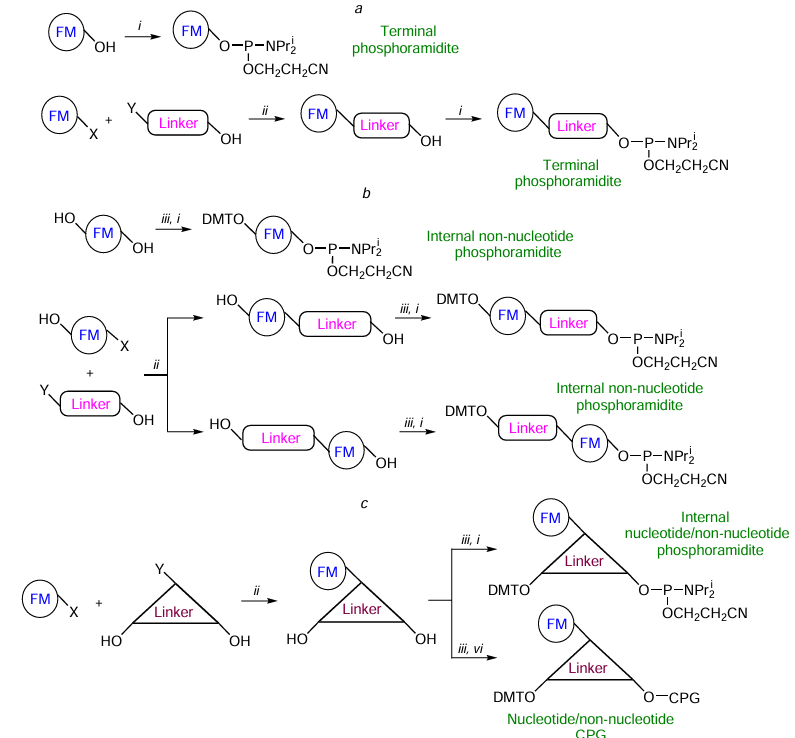
As regards their synthesis, the simplest modifiers are non-nucleotide terminal phosphoramidites. These compounds are usually synthesized in one or two steps using FMs containing only one hydroxyl group or reaction centre (designated by Х in Fig. 4a) capable of reacting with simple bifunctional hydroxyl-containing molecules (usually amino alcohols or diols).
Linear internal modifiers can be prepared using either dihydroxyl- or hydroxyl-containing FMs with an additional reaction centre Х in combination with amino alcohols or diols (see Fig. 4b). Branched internal modifiers or functionalized solid supports are usually synthesized starting from FMs that contain the reaction centre Х (see Fig. 4c). In the latter case, nucleosides, amino diols, or triols can act as linkers.
It can be seen from Fig. 4 that the major differences between the modifiers are related to the linker (nucleotide or non-nucleotide) that connects the phosphoramidite group or the solid support to FM. The linker affects the stability of the resulting conjugate, the spatial position of FM, and the functional performance as a whole. For example, dye and quencher phosphoramidites often have special linkers for the optimal arrangement of these labels in the oligonucleotide sequence. Nucleotide linkers mimic the structure of nucleotide units, which is important for preserving the physicochemical properties of DNA or RNA, while non-nucleotide linkers provide additional flexibility and a wider range of modification options. Hence, modifiers are multicomponent systems in which not only the chemical nature of the modifying groups is important, but also the linker structure, which ensures proper attachment and the necessary functionality of FMs (see Fig. 2 where the key sites for FM introduction are indicated by coloured arrows).
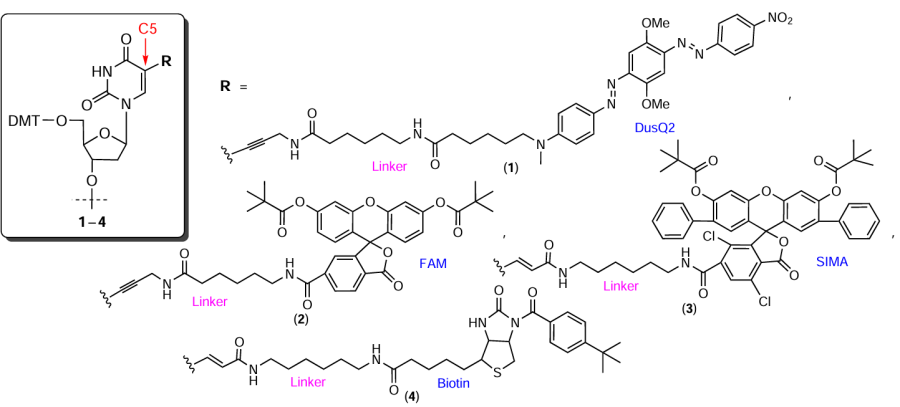
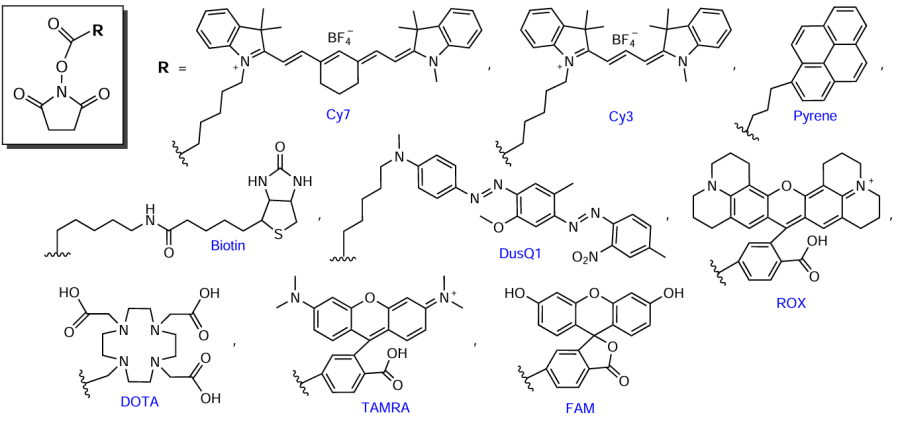
Considering commercial modification reagents, most often, they are nucleotide derivatives or non-nucleotide inserts containing pyrene, cholesterol, α-tocopherol, psoralen, and biotin or quenchers and dyes (FAM, JOE, TAMRA, Cy, HEX, etc.).* Various manufacturers offer their own unique linkers differing in the chemical structure and the mode of attachment to the oligonucleotide, which allows tailoring of the synthetic protocol to specific applications. For example, the use of modifiers 1 – 4 containing FM residues in heterocyclic bases allows effective targeting of the introduced functional groups to NAs or proteins (Fig. 5).** The presence of long linkers in the macromolecule between the heterocyclic base and FM minimizes the effect of oligonucleotide modification on the complementary Watson – Crick interactions and on the spatial structure of the resulting complexes.
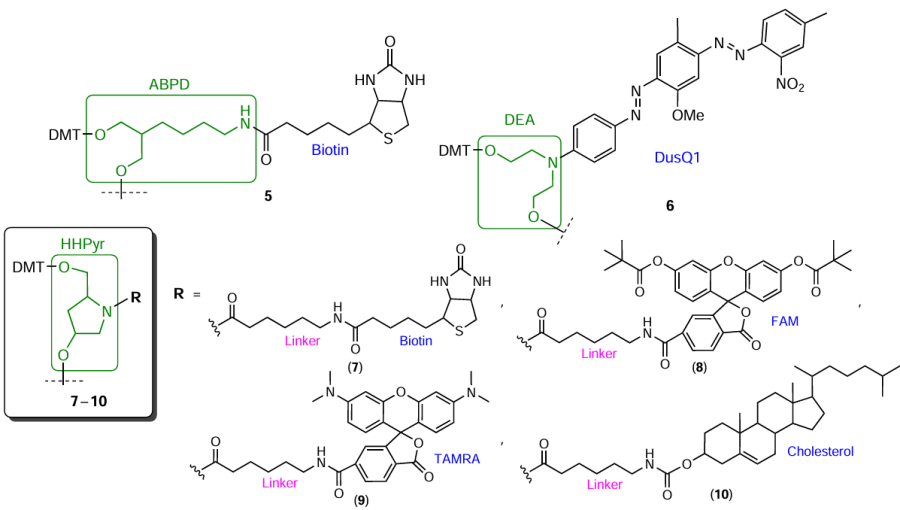
For commercial non-nucleotide FM-containing phosphoramidites and/or solid supports, quite popular are groups based on aminodiols, which allow the preparation of both terminal and internal modifying reagents. In particular, 2-(4-aminobutyl)propane-1,3-diol (ABPD), diethanolamine (DEA), and 4-hydroxy-2-(hydroxymethyl)pyrrolidine (HHPyr, hydroxyprolinol) proved to be effective as structural units providing both successive elongation of the oligomer chain (owing to the presence of two hydroxyl groups) and FM attachment to the aliphatic amino group either directly (Fig. 6, compounds 5, 6) or via an additional linker (compounds 7 – 10).
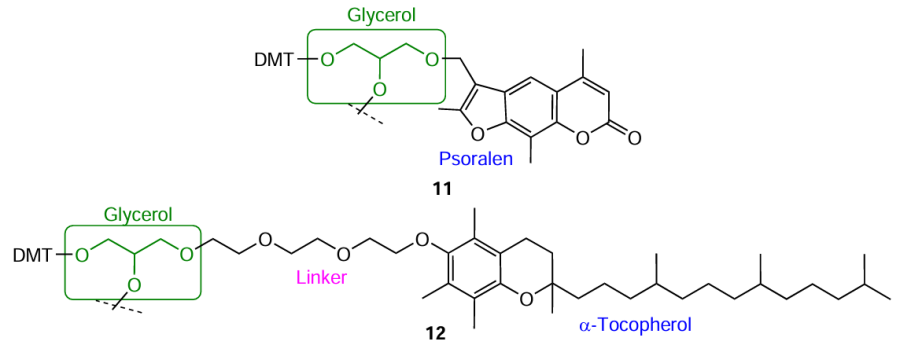
Glycerol and glycidol (2,3-epoxypropan-1-ol) in combination with glycols are used for the production of commercial modifiers 11 and 12, which can be incorporated into any position of the oligonucleotide chain (Fig. 7).
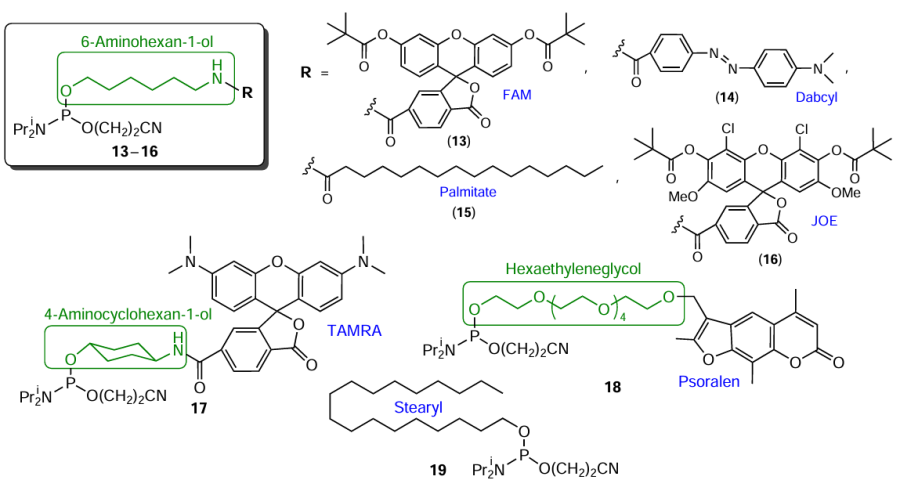
Terminal modifiers are phosphoramidites that preclude further elongation of the oligonucleotide chain. These compounds are obtained using amino alcohols or diols as linkers (Fig. 8, compounds 13 – 18), or by converting FMs (those containing a hydroxyl group in the molecule) to 5'-terminal modifiers for automated synthesis (e.g., compound 19).
Fig. 5 show examples of compounds most of which are non-nucleotide synthons that belong to the class of fluorescent dyes (quenchers). Thus, approximately one hundred most commonly used modifiers meant for the synthesis of oligonucleotide conjugates of various types and functions are commercially available.
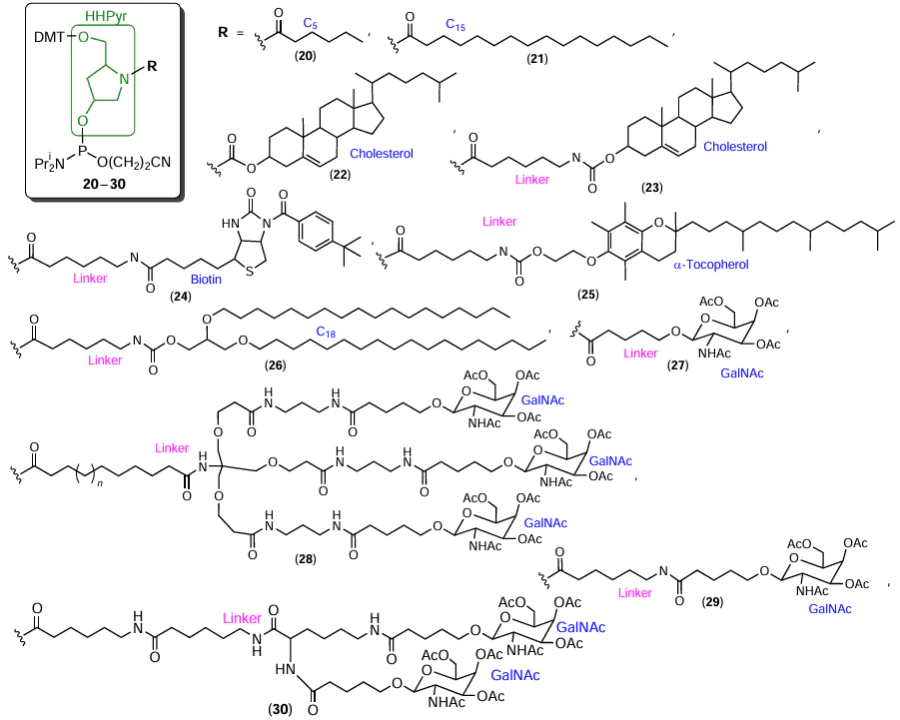
In addition, there are publications describing the preparation and use of a number of lab-made modifiers for automated synthesis. A highly popular synthon is hydroxyprolinol (formed upon the reduction of the corresponding amino acid, L-hydroxyproline), which allows for convenient introduction of FM residues into various positions of oligonucleotides. The use of HHPyr to prepare modified phosphoramidites and solid supports is described in detail by Tatulchenkov et al.[86] In particular, the authors prepared biotin-containing modifiers (see, for example, compound 7 in Fig. 6). The synthesis of phosphoramidites containing caproic (C5) and palmitic (C15) acid moieties (Fig. 9, compounds 20, 21) is described by Tanaka et al.[87] The cholesterol residue can be attached to hydroxyprolinol either directly[46] or via aminocaproic acid-based linker[47] (compounds 22, 23). The synthesis of modifiers containing biotin, α-tocopherol, aliphatic C18 chain (compounds 24 – 26) has been patented.[88] Hydroxyprolinol modifiers containing one or a few N-acetylgalactosamine residues (compounds 27 – 30) have been described in a number of publications.[89-99]
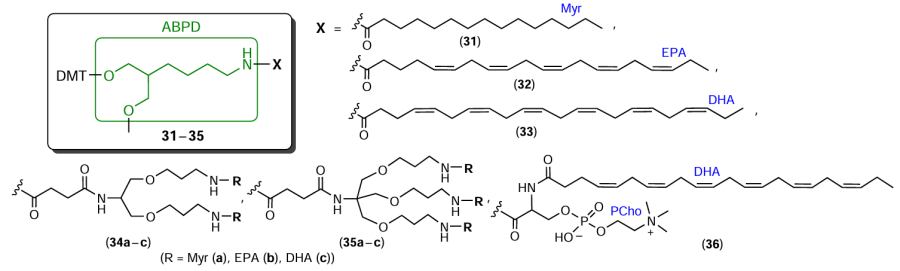
The linear aminodiol ABPD was used[100] to prepare a series of reagents 31 – 35 containing one, two, or three myristic (Myr), eicosapentanoic (EPA), or docosahexaenoic (DHA) acid residues (Fig. 10). Subsequently, the same authors described compound 36, which contains DHA and phosphocholine (PCho) moieties.[101]
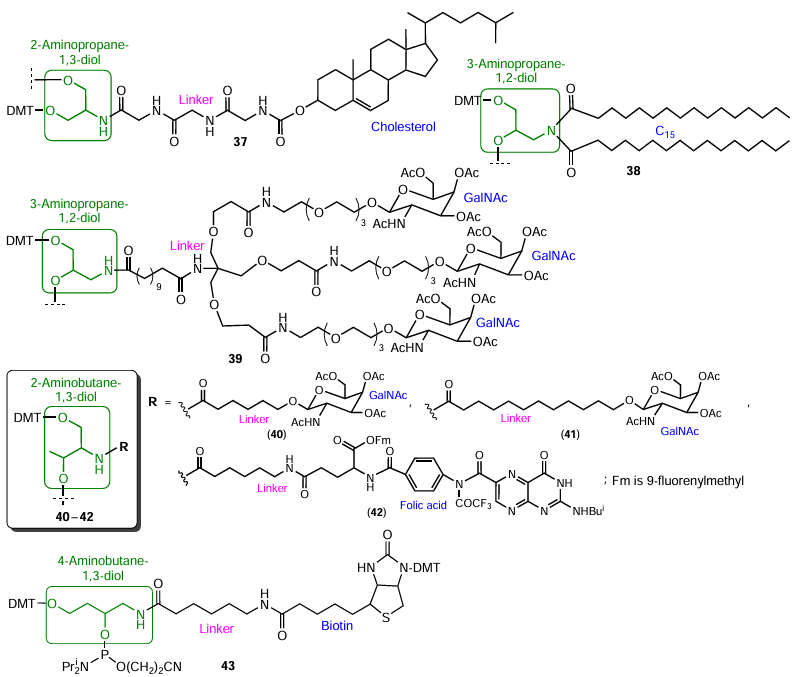
A cholesterol-containing reagent can be prepared, for example, using a short aminodiol, serinol (2-aminopropane-1,3-diol), in combination with triglycine linker[102] (Fig. 11, compound 37). Structural analogues of serinol, 3-aminopropane-1,2-diol and D-threoninol (2-aminobutane-1,3-diol), were used to obtain modifiers containing several palmitic acid (C15) (compound 38)[103] or N-acetylgalactosamine (GalNAc) residues (compounds 39 – 41).[104][105] The use of D-threoninol[105] also resulted in the synthesis of folic acid phosphoramidite 42. Using 4-aminobutane-1,3-diol formed upon the reduction of the carboxyl group in 4-amino-3-hydroxybutanoic acid to hydroxymethyl group, Neuner[106] prepared biotin phosphoramidite 43.
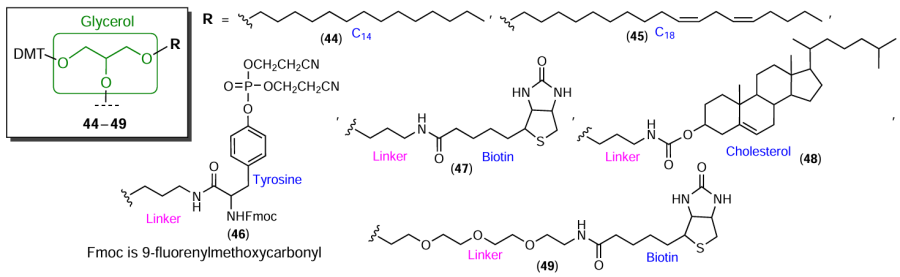
Glycerol is used equally often for the laboratory synthesis of modification reagents. Commercially available solketal (glycerol in which two OH groups are isopropylidene-protected) and 1-bromotetradecane or octadeca-1,13-dienylmethanesulfonate were converted to glycerol phosphoramidites and solid supports containing hydrophobic C14- and C18-aliphatic chains[107][108] (Fig. 12, compounds 44, 45). Using 3-aminopropylsolketal, Misiura et al.[109] prepared tyrosine and biotin O-bis(2-cyanoethyl) phosphate phosphoramidite derivative (compounds 46 and 47). In this case, like in the synthesis of cholesterol-modified solid support and phosphoramidite modifier[102][110] (e.g., compound 48), 3-aminopropanol served as the linker. The biotin molecule was converted to phosphoramidite 49 based on tetraethylene glycol and 1-dimethoxytrityl-2,3-epoxypropanol.[111]
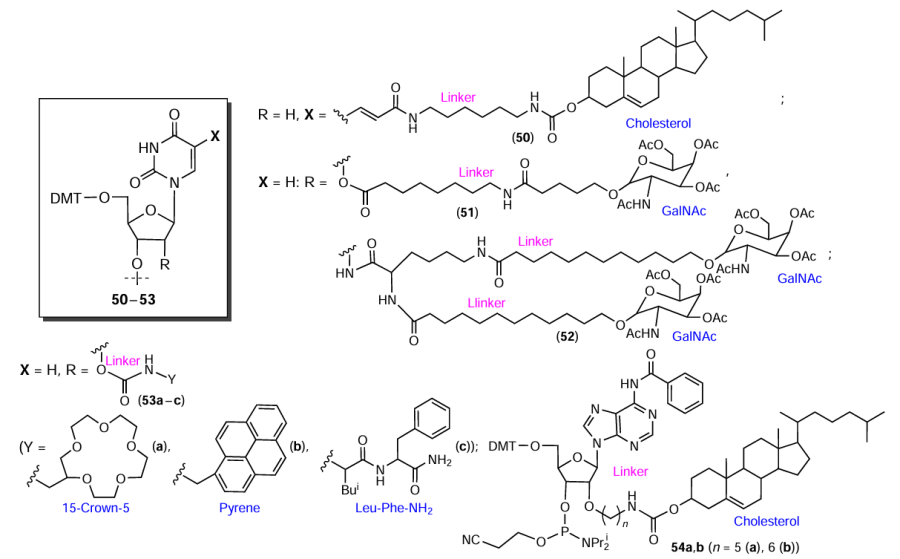
In the synthesis of nucleotide modifiers, various FMs are usually attached to the C5 position of uridine[88] (e.g., compound 50 in Fig. 13) or 2'-position of ribose[94][105][112-114] (compounds 51 – 54).
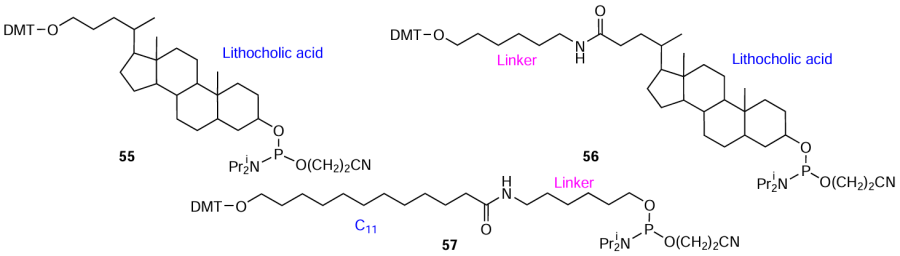
Functional molecules containing at least two reactive groups can be used as non-nucleotide inserts by being directly converted to phosphoramidites suitable for automated synthesis. For example, the reduction of the carboxyl group in lithocholic acid to primary hydroxyl group[115] afforded compound 55 (Fig. 14), while introduction of an amino alcohol linker gave compound 56 (see Fig. 14).[116] Lorenz et al.[115] also synthesized a non-nucleotide insert based on 12-hydroxylauric acid (C11) (compound 57).
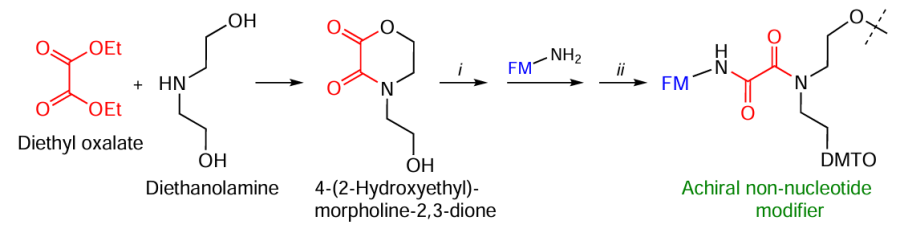
Kupryushkin et al.[117-119] proposed a synthetic route toward cyclic 4-(2-hydroxyethyl)morpholine-2,3-dione, a versatile platform for the preparation of non-nucleotide terminal and/or internal modifiers, using diethyl oxalate in combination with diethanolamine (Fig. 15). The successive introduction of the dimethoxytrityl protecting group, opening of the lactone ring in the presence of FM containing an aliphatic amino group (see below Section 2.2.1), and phosphitylation/attachment to solid support furnished a series of standardized achiral non-nucleotide modifiers the functional properties of which are determined by the FM nature.
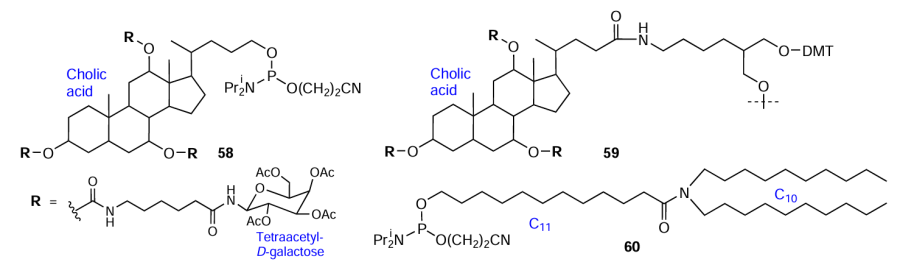
A few FM residues can be combined in one modifier molecule.[115][120] The combination of cholic acid and D-galactose residues made it possible to prepare both 5'- and 3'-terminal as well as internal modifiers 58, 59 (Fig. 16).[120] Lorenz et al.[115] designed compound 60, a structural analogues of membrane lipids, using a combination of 12-hydroxylauric acid (C11) and di-n-decylamine (C10) (see Fig. 16).
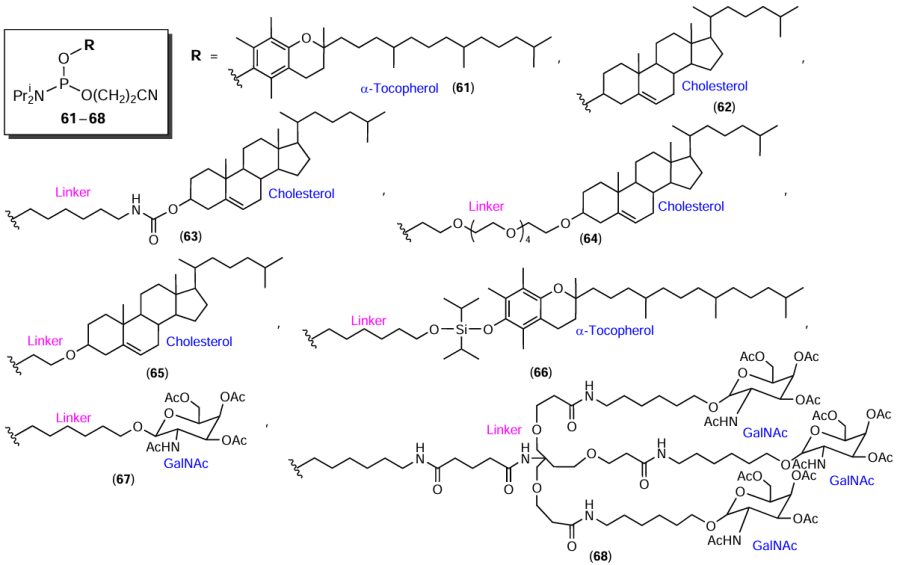
The presence of primary and secondary hydroxyl groups in FMs normally allows for the preparation of terminal phosphoramidites for the automated synthesis of 5′-сonjugated оligonucleotides. Compounds 61 and 62 are modifiers containing α-tocopherol[121-123] and cholesterol[121][124-126] residues (Fig. 17).
In addition, FMs can be modified with amino alcohols or diols (in particular, glycols) to extend the linker between FM and the oligonucleotide. For example, the same Figure shows examples of terminal linker-containing phosphoramidites 63 – 68 with cholesterol, α-tocopherol, and N-acetylgalactosamine residues.[95][127-130]
The part of the review dealing with the pre-synthetic approach to the preparation of oligonucleotide conjugates with various FM residues can be summarized by the conclusion that commercially available modifiers are usually those containing various, partially standardized residues of dyes, quenchers, or hydrophobiс agents, which determine the biospecific behaviour of the molecules. Lab-made modifiers are mainly FMs that ensure the oligonucleotide delivery owing to their hydrophobicity or ability to bind to a particular receptor.
* Generally accepted designations for commercial dyes are used; examples of structural moieties of some dyes are shown in the Figures.
** The structures presented here and below are examples of modifiers for automated oligonucleotide synthesis containing FM moieties or reactive groups and available both as commercial and lab-made chemicals (the dashed line indicates a phosphoramidite group or a solid support; the wavy line indicates the site of substituent attachment).
4. Post-synthetic approach: integrated strategies for the synthesis of conjugates
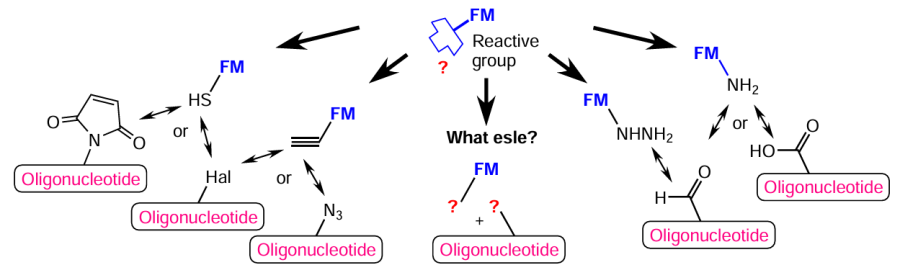
Despite the convenience and diversity of commercially available modifiers that enable the synthesis of conjugates with diverse functional properties, the pre-synthetic approach still does not make it possible to fully address all challenges faced by researchers. Often, it is necessary, first, to obtain, by the automated synthesis, an oligonucleotide derivative that can further be converted to FM-conjugate. The post-synthetic approach to conjugation of oligonucleotides, both on a solid support and in solution (see Table 1), is utilized in those cases if FM conjugated with oligonucleotide is labile during chemical reactions involved in the oligonucleotide synthetic cycle and/or final deprotection procedures (see Fig. 1). Synthetic protocols that take into account the type and position of reactive groups in both components (oligonucleotide and FM) are elaborated for each particular conjugate (Fig. 18). While planning a synthetic protocol, one should proceed, first of all, from the structural characteristics of FM that determine the optimal approaches for potential modification. Moreover, it should be borne in mind that the most appropriate preliminary modification of oligonucleotides is that ensuring the highest yield of the corresponding DNA and RNA derivative and the highest conversion of this derivative in the reaction with activated FM.
The most commonly used reactive groups for post-synthetic conjugation are carboxyl, hydroxyl, aldehyde, amine, hydrazide, azide, alkyne, phosphate, and phosphite groups and halogen atoms. The following combinations of reactive groups that enable successful conjugation of oligonucleotides and FM can be distinguished: amine + carboxyl, amine + hydroxyl, amine + halide, amine + aldehyde, hydrazide + aldehyde, alkyne + azide, alkyne + halide, thiol + halide, thiol + maleimide, thiol + thiol, amine + phosphate, amine + phosphite, and phosphite + azide. In most cases, the groups in the pairs are complementary, which allows for orthogonal pair arrangement in the oligonucleotide – FM system to facilitate interaction between the groups for the formation of the target compound. The post-synthetic approach is popular, since it provides efficient synthesis of oligonucleotide conjugates with a variety of functional groups both during the solid-phase synthesis stage and after deprotection and isolation of the final product (e.g.[131-136]).
It is also important that in the case of multifunctional oligomers carrying several non-nucleoside biologically significant residues, the method for their preparation should be based on either separate conditions of modification reactions during different stages of oligomer synthesis (combination of the pre- and post-synthetic approaches) or the use of an integrated post-synthetic strategy (combination of several types of reactive groups). Examples of such combinations of chemically orthogonal groups that react under strictly specific conditions include carboxyl and amino groups (amide bond formation) and alkynyl and azide groups (click reaction).
While planning the synthesis of an oligonucleotide – FM conjugate, one should take into account structural organization of the components. It is important to understand which groups in the oligonucleotide and FM can serve as conjugation sites, while preserving their biological functions and providing the appearance of specific properties of the combined macromolecule.
4.1. Preparation of conjugates using amino and carboxyl components
The reaction between two compounds, one containing an amino group and the other containing a carboxyl group, resulting the formation of the covalent amide bond is widely used in the synthesis of oligonucleotide conjugates with FMs (Fig. 19).
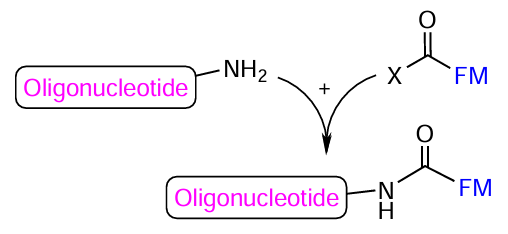
A classic approach to the formation of NA conjugates is the acylation of oligonucleotides containing an aliphatic amino group in various positions of the oligonucleotide chain with activated carboxyl-FMs (Fig. 20). This type of conjugation is often used. For example, this approach is effective for the post-synthetic preparation of oligonucleotide derivatives with cyanine dyes Cy7 and Cy7.5 where FM is unstable during automated synthesis and NA deprotection procedure.
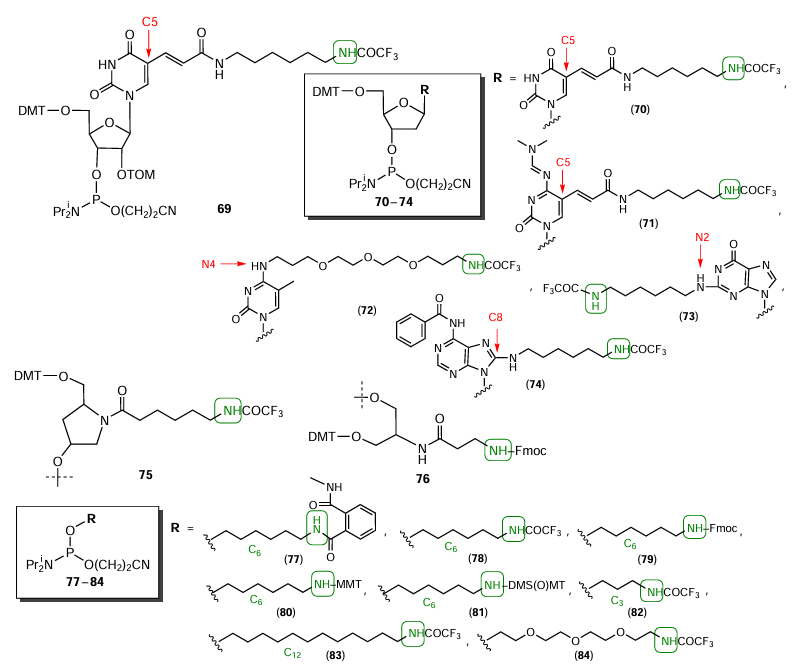
Fig. 21 shows typical examples of commercially available phosphoramidites or solid supports that allow the introduction of aliphatic amino groups into any position of the oligonucleotide chain. As the modification sites of nucleosides, it is convenient to use the C(5) positions of pyrimidines of ribo (compound 69) or deoxyribo derivatives (compounds 70 – 71), the N(4) position of cytidine (compound 72), and N(2) and C(8) positions of purines (compounds 73 – 74). Amino modifiers 75 and 76 based on hydroxyprolinol and serinol, respectively, also make it possible to introduce an amino group as a part of non-nucleotide insert into any position of the oligonucleotide chain. Commercial terminal amino modifiers 77 – 83 based on amino alcohols containing 3, 6, or 12 carbon atoms and compound 84 containing a glycol moiety have been offered. For convenience of researchers, apart from standard alkali-labile protecting groups (phthalimide and trifluoroacetyl groups in compounds 77 and 78), other groups have been proposed such as monomethoxytrityl (MMТ in compound 80) and 4,4'-dimethoxy-4''-sulfonyltrityl [DMS(O)MT in compound 81] acid-labile protecting groups and fluorenylmethoxycarbonyl group (Fmoc in compound 79) that can be selectively removed under the action of bases (e.g., piperidine). Thus, the reaction potential of these groups can be implemented, with most other protective groups in the oligonucleotide being intact. The use of protecting groups with different stabilities makes it possible to selectively deprotect the amino group with other protecting groups being present in the oligonucleotide, or, conversely, to preserve the protected amino group after deprotection of the oligomer before the modification.
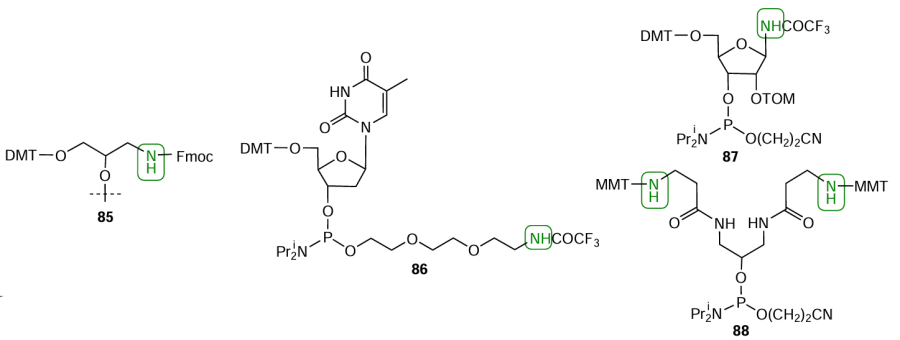
The simplest and least costly way of introduction of amino group as a non-nucleotide insert into any oligonucleotide position in the laboratory synthesis of amino modifiers was proposed by Nelson et al.[137][138] The method includes the use of 3-aminopropyl-1,2-diol as a modification scaffold (Fig. 22, compound 85). Using 2-[2-(2-chloroethoxy)ethoxy]ethanol, Tabatadze et al.[139] obtained thymidine phosphoramidite 86 modified at the phosphorus atom. Rydzik et al.[140] prepared ribofuranose analogue based on 1-amino-2,3-dihydroxy-4-(hydroxymethyl)cyclopentane, namely, 1'-aminocarbocyclic ribophosphoramidite 87 containing protected amino group at the C(1') position. Lab-made 5'-terminated bis-amino modifier 88 was prepared[141] using β-alanine and 1,3-diaminopropan-2-ol.
The approaches that can serve to introduce amino groups in the ribose and heterocyclic base moieties or in internucleoside phosphate group are described in reviews.[142][143]
In the acylation reactions for the synthesis of conjugates, a special place belongs to activated N-hydroxysuccinimide (NHS) esters of FMs, as they can provide a high conversion of amino-containing oligonucleotide to the target conjugate even when water serves as a solvent (see examples of commercial NHS esters in Fig. 23). The reaction between NHS-esters of FM and amino-modified oligonucleotides proceeds under relatively mild conditions in an organic or aqueous organic medium at room temperature within several hours. An important factor for this reaction is pH of the solution, which can vary from 7.5 to 9.5. The optimal pH values for the acylation of aliphatic amino groups in aqueous solutions are in the range of 8.3 – 8.5.[144][145] As a rule, 0.1 M solutions of sodium bicarbonate or sodium tetraborate are used as buffer solutions. Alternatively, it is possible to use 0.1 M phosphate buffer or Tris · HCl-based buffers [Tris is tris(hydroxymethyl)aminomethane]. If the reaction is carried out in a polar aprotic solvent (e.g., DMF or DMSO), a tertiary amine, such as triethylamine, must be added to the reaction mixture in an amount equivalent to the content of the NHS ester.
The reaction shown in Fig. 20 is suitable for the synthesis of a wide range of oligonucleotide – FM conjugates by combining various amine and succinimide components, since commercial manufacturers offer more than 60 types of NHS esters, including derivatives of pyrene, biotin, fluorescent dyes, quenchers, etc.
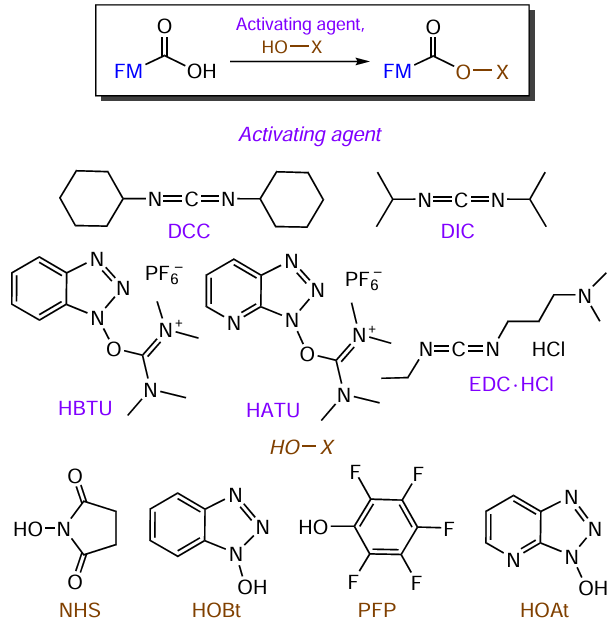
Activated FM esters intended for the modification of oligomers are synthesized using carbodiimides that react with carboxylic acids to give an intermediate, highly reactive O-acylisourea derivative. Currently, N,N'-dicyclohexylcarbodiimide (DCC), N,N'-diisopropylcarbodiimide (DIC), and water-soluble 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide (EDC · HCl) are used most often to activate carboxyl groups[146] in FMs toward the synthesis of oligonucleotide conjugates (Fig. 24).
N,N'-Dicyclohexylcarbodiimide was one of the first carbodiimides to be used as activating reagent for the formation of the C(O) – NH amide bond.[147][148] This reagent is readily soluble in many organic solvents (dichloromethane, tetrahydrofuran, acetonitrile, dimethylformamide, etc.), but is insoluble in aqueous solutions. While using DCC, it should be borne in mind that N,N'-dicyclohexylurea formed as a by-product is insoluble in most organic solvents. Therefore, this reagent is incompatible with the heterogeneous method of conjugate synthesis. N,N'-Diisopropylcarbodiimide widely used in organic synthesis[149] is a colourless liquid suitable for the solid-phase synthesis, while the product of DIC activation of carboxyl group, 1,3-diisopropylurea, is soluble in common organic solvents and is easily removed by washing. Water-soluble EDC · HCl[150][151] effectively activates carboxylic acids in water and aqueous organic solutions.
For the formation of amide bonds involving carbodiimides, additional reagents (co-activators) are also often employed to enhance the reaction efficiency, reduce racemization, prevent the formation of by-products (e.g., N-acylurea), and increase the yield of the target compound (see Fig. 24). By combining carbodiimide and N-hydroxysuccinimide[152][153] or pentafluorophenol (PFP),[154] it is possible to obtain NHS- or PFP-activated carboxylic acid esters possessing relatively high storage stability at a reduced temperature when protected from water. Since carbodiimide activation may induce racemization of the activated amino acid,[146] it is reasonable to perform activation by compounds such as 1-hydroxybenzotriazole (HOBt)[155] and 1-hydroxy-7-aza-benzotriazole (HOAt).[156] They prevent this transformation and provide high reactivity for the corresponding ester, which subsequently reacts with an aliphatic amine to form an amide bond. To date, a broad range of activating agents has been developed using these compounds, including 2-(1H-benzotriazol-1-yl)-1,1,3,3-tetramethyluronium hexafluorophosphate (HBTU)[157-159] and its 7-aza-analogue (HATU),[159][160] which are characterized by a low degree of racemization.
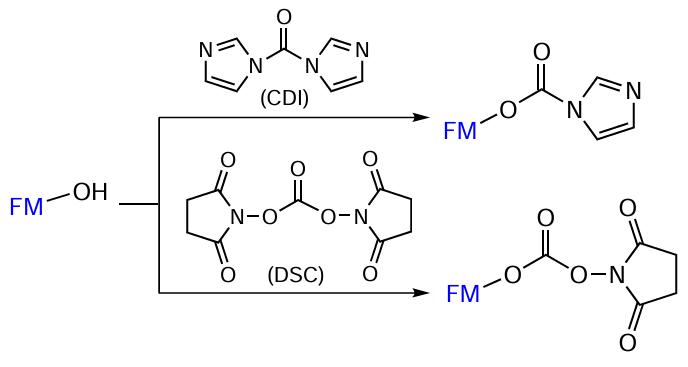
The oxidation of aldehyde group in FMs to carboxyl group is performed using a mild oxidant, Tollens’ reagent (ammonia solution of silver oxide).[161] Compounds containing hydroxyl groups can be converted to activated derivatives capable of reacting with the amine component of oligonucleotide using, for example, carbonyldiimidazole (CDI)[162-167] or N,N-disuccinimidyl carbonate (DSC)[168-173] (Fig. 25).
The reactions shown in Fig. 24 and Fig. 25 are also fast and efficient; moreover, activated FMs do not require purification and can be introduced in situ into reactions with amino-containing oligonucleotides if equivalent amounts of activating agents (carbodiimides, CDI, and DSC) and carboxyl/hydroxyl-containing FMs are used.
The inverted type of reaction between carboxyl-modified oligonucleotides and amino-FMs is frequently utilized for the preparation of conjugates according to the post-synthetic heterogeneous synthesis strategy (Fig. 26).
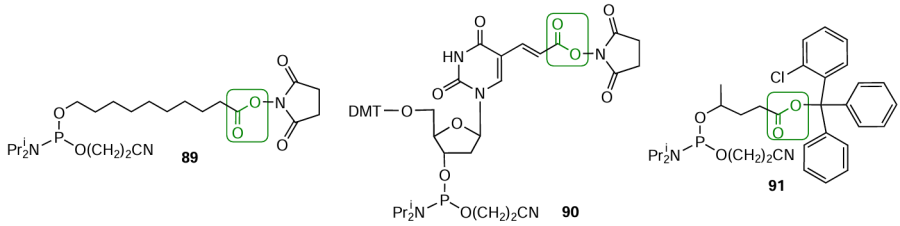
Commercial carboxyl-modified phosphoramidites (Fig. 27) suitable for automated oligonucleotide synthesis have been proposed for modification of both the 5'-end and the internal part of the oligonucleotide chain. Modifiers 89 and 90 contain activated ester moieties that tend to form amide bonds upon reactions with aliphatic amines. The 2-chlorotrityl group in 5'-carboxy modifier 91 is a protecting group removed during the standard acid-catalyzed deprotection to give free carboxyl group in a fully protected immobilized oligonucleotide. After activation with, for example, N-hydroxysuccinimide in the presence of carbodiimides, this carboxyl group is able to react with amino-FMs.
A review by Kachalova et al.[174] addresses the methods of synthesis of oligonucleotides containing a carboxyl group in any position of the oligomer chain: the 5'- and/or 3'-end or inside the chain.
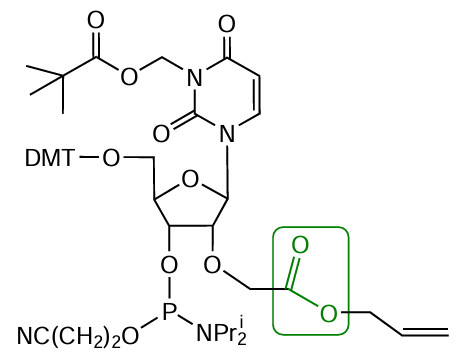
For instance, Kachalova et al.[175] described a modifier with a protected carboxyl group at the 2'-position of the ribose (Fig. 28). The O-allyl protection is stable during the solid-phase assembly of oligonucleotides, but is readily removed by treatment with Pd(PPh3)4 in morpholine. The oligoribonucleotides with a 2'-O-carboxymethyl group that were obtained using this modifier were effectively conjugated via heterogeneous reactions with histamine, spermine, aminopyrene, pyrenemethylamine, and short peptides.
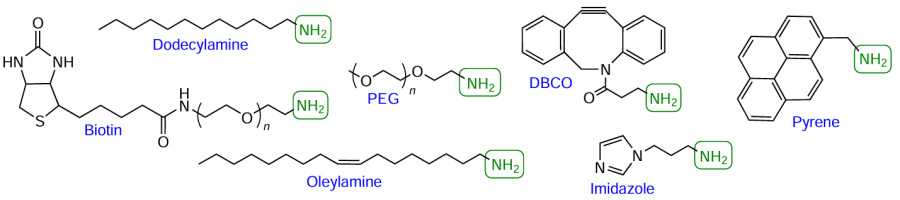
As examples of commercially available amino-FMs, consider polyoxyethyleneamine biotin derivative, dodecylamine, oleylamine, O-(2-aminoethyl)-O'-methyl-substituted polyethylene glycol (PEG), dibenzocyclooctyne-amine (DBCO-amine), 1-(3-aminopropyl)imidazole, and pyrenemethylamine (Fig. 29).
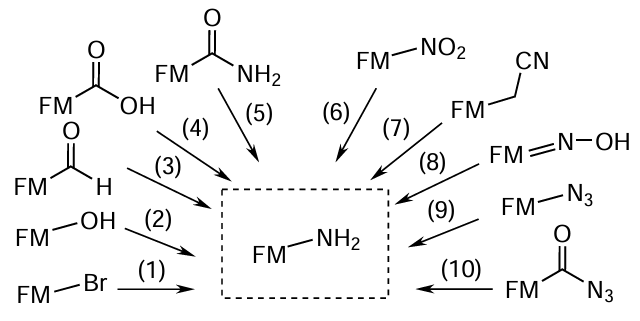
Among the standard laboratory methods for the preparation of amino-FMs, note the following processes[176] (Fig. 30):
— direct and indirect alkylation (1): reaction of primary and secondary alkyl halides with ammonia (direct alkylation, Hofmann reaction[177]); alkylation of potassium phthalimide with alkyl halides to form N-alkylphthalimide followed by conversion to a primary amine on treatment with hydrazine hydrate in an alcohol solution (indirect alkylation, Gabriel synthesis of primary amines[178]);
— catalytic amination of alcohols: passing a mixture of alcohol and ammonia vapours through heated oxides of divalent and trivalent metals (most often, aluminium and thorium), which act as catalysts (2);[179]
— reductive amination of carbonyl compounds: treatment of aldehydes with ammonia and simultaneous reduction with hydrogen [involving the intermediate formation of imines (Schiff bases), which react with hydrogen in the presence of catalysts for heterogeneous hydrogenation] (3);[180][181]
— reductive amination of carboxyl compounds by the Schmidt reaction according to which carboxylic acids react with hydrazoic acid in acidic medium; the reaction involves the intermediate formation of isocyanate, which is converted to amine with the release of molecular nitrogen and CO2 (4);[182][183]
— Hofmann degradation of acid amides: conversion of amides on treatment with alkali metal hypochlorites (hypobromites) or halogens in alkaline medium giving primary amines containing one carbon atom less than the original amide (5);[184]
— reduction of nitro compounds by catalytic hydrogenation [catalyzed by silica-supported copper; nickel in combination with vanadium and aluminium oxides; platinum(IV) oxide; Raney nickel] or by treatment with reducing agents: metals (lead, zinc, or iron chips) or metal salts in the presence of acids and water; reduction of nitro groups in aromatic systems using alkali metal hydrosulfides and disulfides (6);[185-187]
— catalytic hydrogenation of nitriles with lithium aluminium hydride in diethyl ether or sodium borohydride (7);[188][189]
— reduction of oximes with lithium aluminium hydride or sodium cyanoborohydride (8);[190]
— reaction of azides with lithium aluminium hydride[191] or hydrogen iodide (9);[192]
— thermal decomposition of carboxylic acid azides (Curtius rearrangement[193-198]): rearrangement of acyl azides to isocyanates on heating, with the release of nitrogen gas and the subsequent decarboxylation of intermediate carbamic acids resulting from the reaction of isocyanate with water (10).
The history of investigation of the amide bond C(O) – NH (particularly, peptide bond) dates back to the late 19th century when German chemist E.Fischer (cited from Jaradat[199]) proposed the term ‘peptide’ and a general method for laboratory synthesis of peptides using amino acid chlorides. This discovery not only formed the basis for further studies of peptides, but later, also made it possible to apply all the accumulated experience to the production of oligonucleotide conjugates with various FMs (in particular, with peptides[200-202]). This discovery resulted in the development of peptide nucleic acids (PNAs), electrically neutral synthetic DNA analogues in which the sugar – phosphate backbone is replaced by a pseudopeptide backbone.[203-207]
4.2. Preparation of conjugates using amino and hydroxyl components
The reactions involving amino and hydroxyl groups are used to prepare oligonucleotide conjugates only in the case of heterogeneous processes.
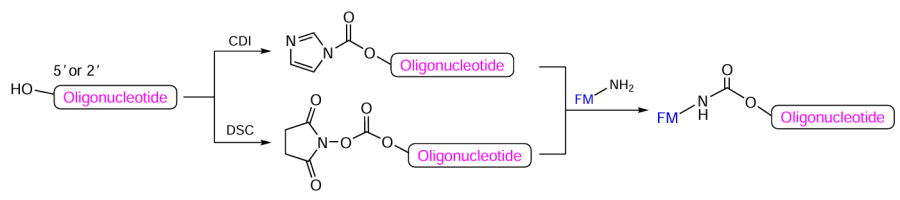
For the preparation of oligonucleotide 5'- and 2'-conjugates, it is possible to activate the hydroxyl group in the specified position of the protected oligonucleotide attached to a support (Fig. 31). Oligonucleotide derivatives activated on treatment with CDI or DSC can further react with molecules that contain primary or secondary amino groups under relatively mild conditions.
In the case of 5'-conjugates, the initial 5'-hydroxyl-containing protected oligonucleotide immobilized on a support is produced immediately after automated synthesis and final detritylation of the resulting chain (Fig. 32). Using this method, it is possible to fabricate a broad range of oligonucleotide 5'-conjugates, for example, with EPR spin labels, lipophilic compounds, vitamins, fluorophores, etc.[208-219]
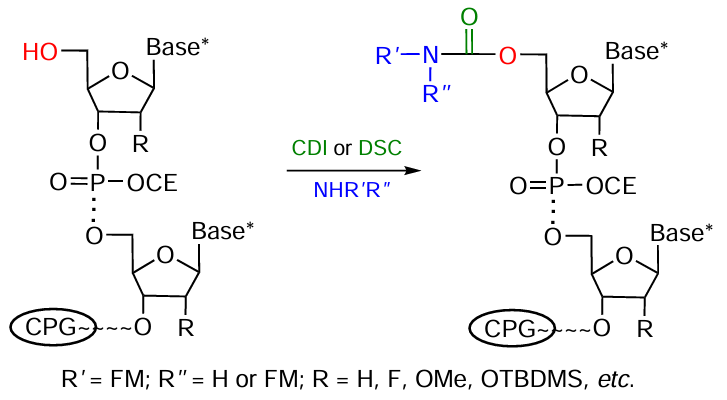
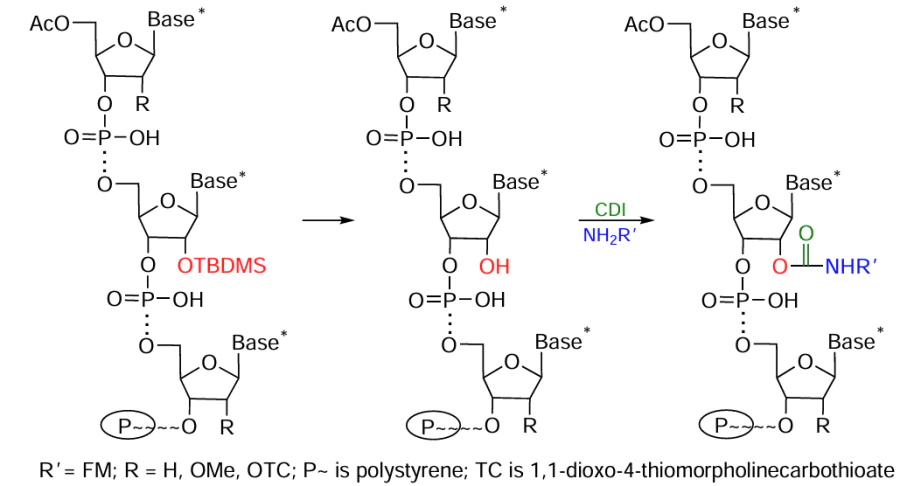
An efficient method for the preparation of oligonucleotides of any type modified at the 2'-position of the ribose residue was proposed by Krasheninina et al.[220] It is based on the use of the 2'-O-ТC-protecting group labile in the presence of alkalis and the 2'-O-TBDMS group that is selectively removed under the action of fluoride ions. The targeted deprotection of 2'-hydroxyl group in the protected immobilized oligomer enables activation of this group and the subsequent addition of amino-FM (Fig. 33). It is noteworthy that the synthesis is carried out on polystyrene supports, which are inert toward the reagents used to remove the TBDMS protection. The proposed method is versatile and is suitable for the preparation of a broad range of 2'-conjugates of RNA and RNA/DNA hybrids.
It is important to note that according to this protocol, the FM conjugation with oligonucleotide is achieved via a carbamate bond, which is stable under physiological conditions.
4.3. Preparation of conjugates using alkynyl and azido components
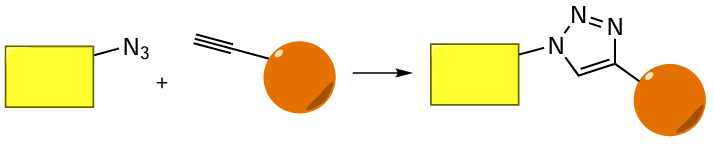
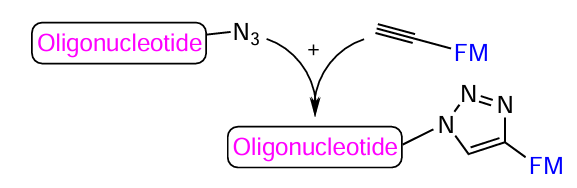
Another popular cross-coupling method meant for the production of oligonucleotide conjugates with various molecules is based on the Huisgen reaction,[221][222] bio-orthogonal Cu-catalyzed azide-alkyne cycloaddition (CuAAC), which regioselectively gives 1,2,3-triazoles (Fig. 34). Catalysis of these reactions by copper(I) cations was reported by M.Meldal[223] and K.B.Sharples[224] research teams working independently of each other. Later, metal-free azide-alkyne cycloaddition method based on the use of cyclooctyne structures was proposed. This protocol of conjugation is called strain-promoted azide – alkyne cycloaddition (SPAAC).[225]The CuAAC and SPAAC processes[226][227] are referred to as click-reactions; for the discovery of these reactions, the authors were awarded the Nobel Prize in Chemistry in 2022.
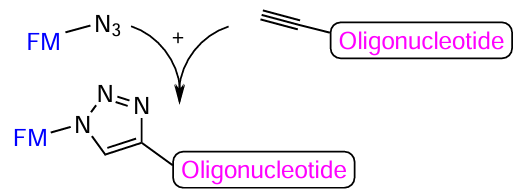
When conjugates are formed by means of a click-reaction, the alkynyl component is, most often, a part of the oligonucleotide (Fig. 35). This is due to the fact that alkynes are stable under the conditions of automated oligonucleotide synthesis, whereas azides tend to be converted under the action of phosphorus(III) compounds (the Staudinger reaction; see Section 4.5 below).[228]
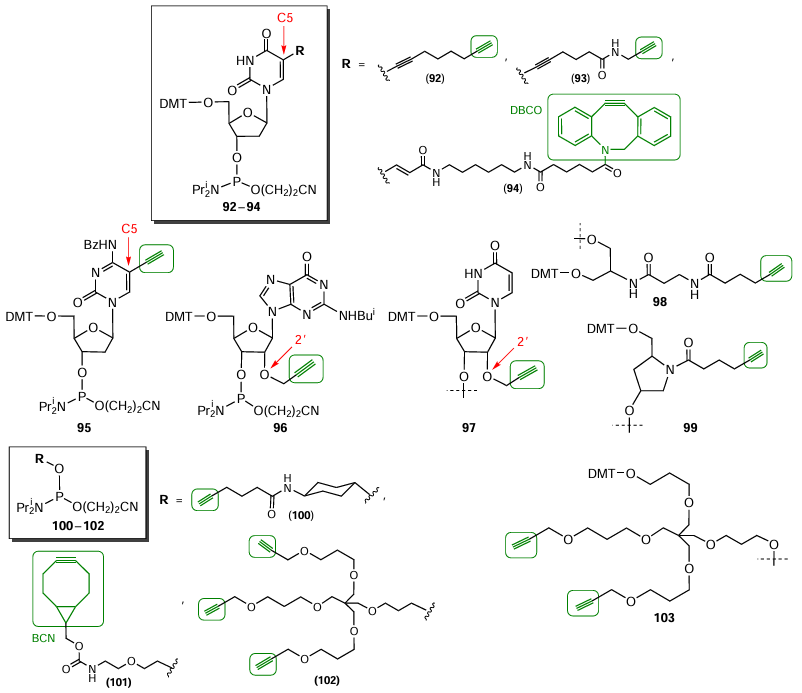
Fig. 36 shows the structures of commercial nucleotide phosphoramidites and specific solid supports in which the triple bond has been introduced into the heterocyclic base (compounds 92 – 94) or into the 2'-position of ribose (compounds 95, 96). Typical non-nucleotide synthons are based on serinol (compound 98), hydroxyprolinol (compound 99), 4-aminocyclohexanol (compound 100), and amino alcohol (compound 101). The use of pentaerythritol [2,2-bis(hydroxymethyl)propane-1,3-diol] makes it possible to prepare both internal and terminal non-nucleotide modifiers 102 and 103 meant for the introduction of two or three alkynyl groups into the oligonucleotide chain. There are commercially available alkynyl modifiers 92 – 93, 95 – 100, and 102 – 103 for copper-catalyzed CuAAC reactions and also strained-ring compounds 94 and 101 containing DBCO and BCN moieties for SPAAC reaction proceeding in the absence of copper cations.
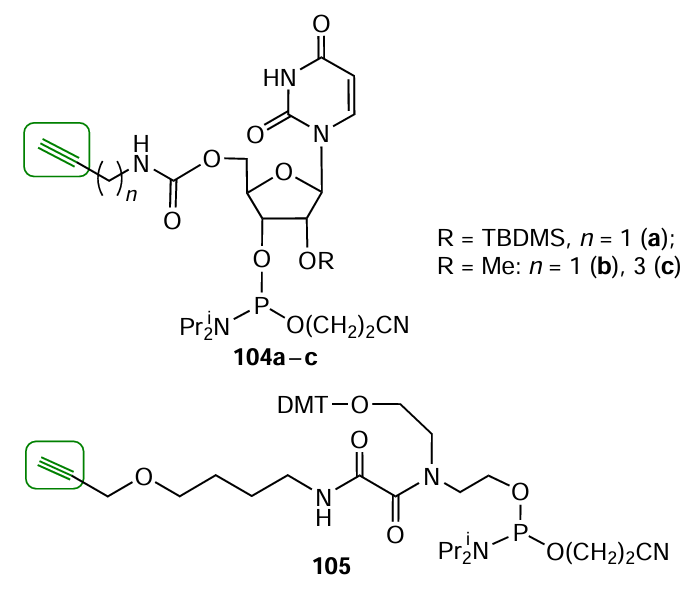
A review by Fantoni et al.[229] describes lab-made alkynyl-modified phosphoramidites or solid supports developed by particular research teams. For example, Yamada et al.[230] reported a series of terminal nucleoside phosphoramidites 104a – c (Fig. 37). The procedure for the synthesis of non-nucleotide synthon 105 based on 4-(2-propynyloxy)butylamine was reported by Kupryushkin et al.[118]
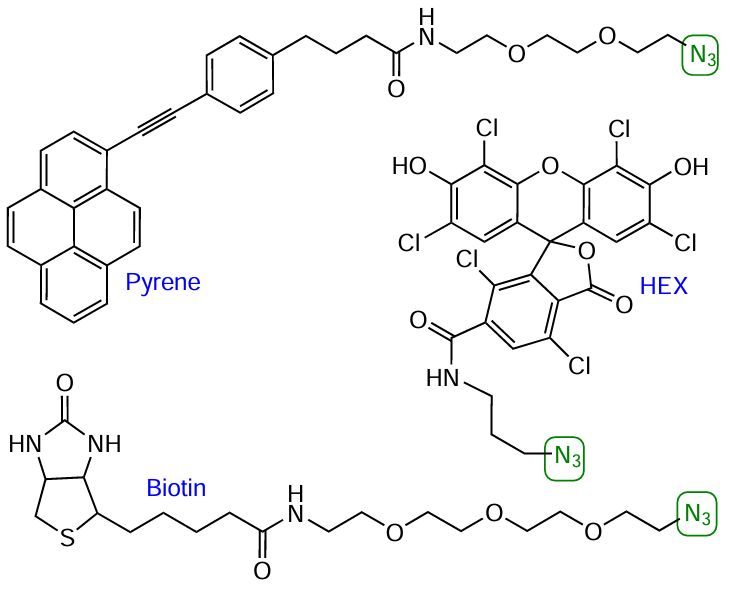
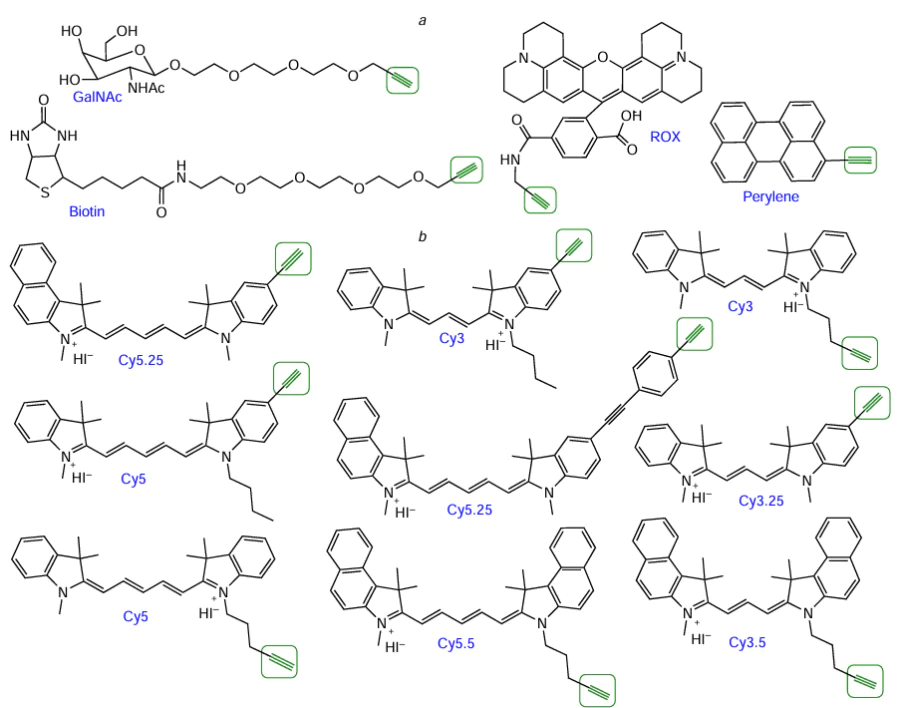
Azido-containing FMs are also widely present in the market, as well as NHS esters of functionalized carboxylic acids (see Section 4.1, Fig. 23). Azide derivatives are available for biotin, pyrene, and quite a few fluorophores: coumarin (AMCA), rhodamine (ROX, R6G, R110, TAMRA, TR, ATT), fluorescein and analogues (FAM, JOE, HEX, TET, VIC, SIMA), cyanine dyes (Cу3, Cу3.5, Cу5, Cу5.5, Cу7, Cу7.5), and boron dipyrromethene dyes (BDP 558/568–BDP 650/665). As an example, Fig. 38 shows the structures of commercial azides based on HЕХ dye, pyrene, and biotin.
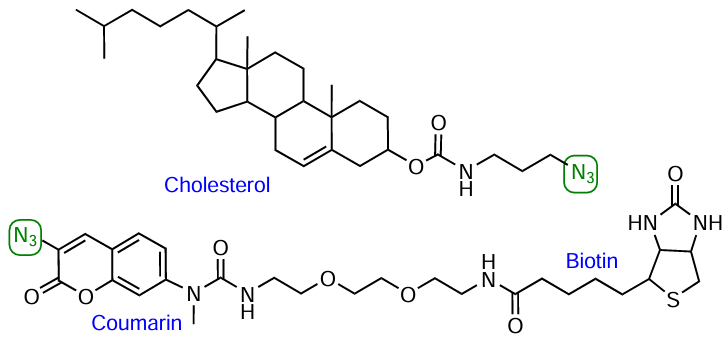
Although commercially available azido-FMs are now used most widely, development of lab-made modifiers is underway. For example, azide cholesterol derivatives[231] and a compound combining coumarin and biotin moieties in the molecule[232] have been reported (Fig. 39).
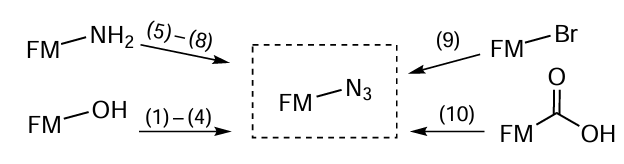
As can be seen from the presented structures, most of the azides are alkyl azides, since the click reactions involving acyl azides are hampered.[233] The target biomolecules containing carboxyl, hydroxyl, or amino group can be easily converted to the corresponding azide derivatives using the following reactions (Fig. 40):[136][197][234-237]
— Mitsunobu reaction of primary and secondary aliphatic alcohols with hydrogen azide, triphenylphosphine, and diethyl azodicarboxylate (1);[238][239]
— treatment of alcohols with 2-azido-1,3-dimethylimidazolinium hexafluorophosphate in the presence of 1,8-diazabicyclo[5.4.0]undec-7-ene (2);[240]
— direct azidation of various alcohols using azidotrimethylsilane in the presence of Amberlyst-15 ion-exchange resin as a catalyst, which can be regenerated and reused (3);[241]
— reaction of alcohols with sodium azide after hydroxyl activation by bis(2,4-dichlorophenyl) phosphate in the presence of 4-(dimethylamino)pyridine (4);[242]
— reaction of a mixture of aromatic amine with tert-butyl nitrite and azidotrimethylsilane under mild conditions (5);[243]
— conversion of amines via diazo transfer reaction involving trifluoromethanesulfonyl azide (CF3SO2N3) as the diazotization agent (6),[244] or imidazole-1-sulfonyl azide hydrochloride,[245][246] a reagent that is easily prepared, commercially available, and stable on storage at a temperature of 4°C (7); or fluorosulfuryl azide in the case of primary amines (8);[247]
— azidation of alkyl halides (typically containing Br or I atom) with sodium azide in polar solvents, such as hexamethylphosphoramide, or using crown ethers (9);[136] [248][249]
— decarboxylating azidation of aliphatic carboxylic acids with tosyl azide or pyridine-3-sulfonyl azide in water under mild conditions using AgNO3 as a catalyst and K2S2O8 as an oxidant (10).[250]
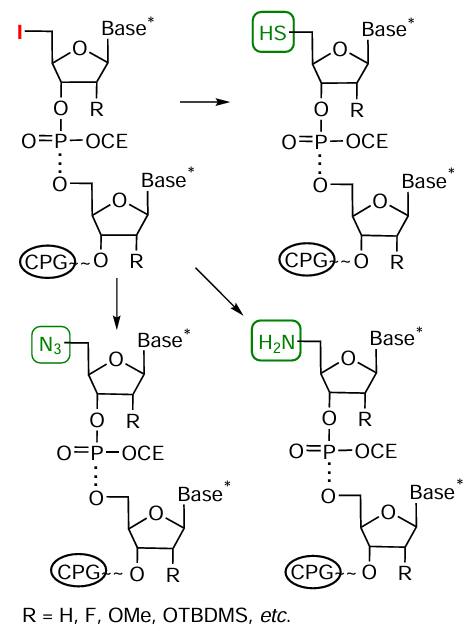
The same conjugates can be prepared using an alternative approach that implies introduction of the azido group into oligonucleotide (Fig. 41).
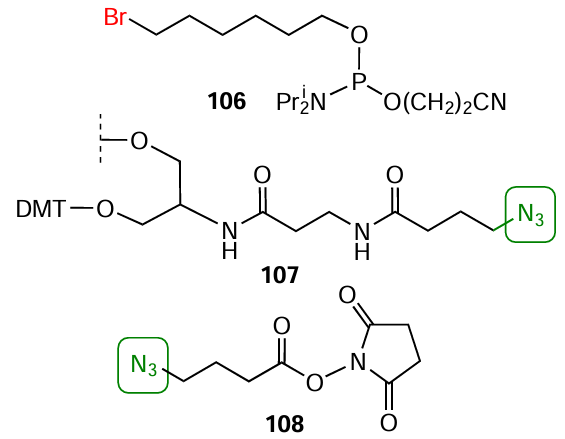
Commercially available bromohexyl phosphoramidite 106 (Fig. 42) is a simple and convenient reagent for the synthesis of 5'-azide-modified oligonucleotide. In this case, the halogen atom substitution by azido group using sodium azide is performed in the final oligonucleotide chain [by analogy with method (9) in Fig. 40]. The introduction of an azido group at the 3'-end of the oligomer chain is carried out, most often, using modified solid support 107 based on serinol (see Fig. 42). Using activated azidobutanoic acid ester 108, the azido group can be introduced by a standard procedure into amino-modified oligonucleotides obtained by automated synthesis using modified nucleotide or non-nucleotide synthons and solid supports (see Fig. 21 and Fig. 22).
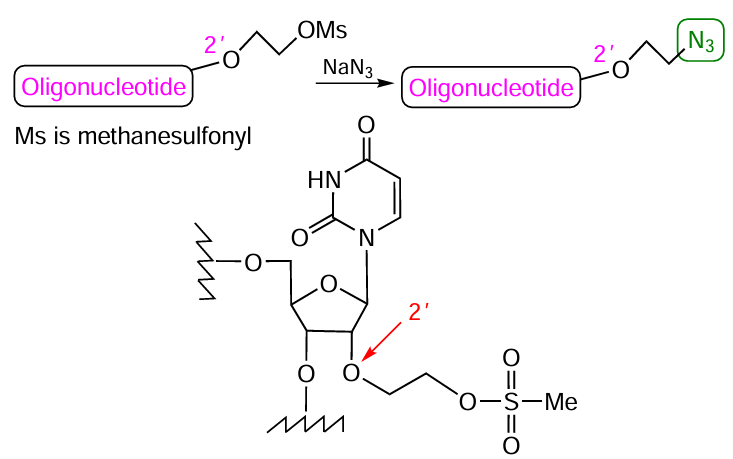
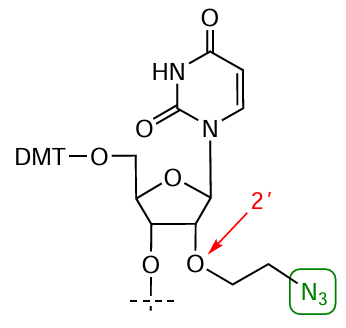
As an example of laboratory synthesis of azido-containing modifiers, note the multistage application of 2'-mesyloxyethyl-containing phosphoramidites, which allow the introduction of the mentioned group in several positions of the synthesized oligonucleotide chain.[251]The substitution of the mesyl group in the oligomer by an azide group on treatment with sodium azide leads to 2'-azidoethyl derivative (Fig. 43).
A solid support containing azido group in the 2'-position or the uridine meant for the synthesis of 2'-O-(2-azidoethyl)-modified RNA[252] is shown in Fig. 44.
Later, it was proposed[253][254] to prepare azido-containing oligonucleotides using diazo transfer reaction induced by fluorosulfuryl azide (FSO2N3). In this case, the initial RNA contains amino group in the heterocyclic base or in the 2'-position of ribose (Fig. 45). It is noteworthy that this reaction gives the corresponding azido-modified RNA in nearly quantitative yield, without affecting the amino groups present in the heterocyclic bases.
A number of reviews[136] [255][256] discuss the strategies for the introduction of alkynyl or azido group into oligonucleotides in more detail.
Among commercially available alkynyl FMs, mention should be made of biotin, N-acetylgalactosamine, perylene, and ROX dye derivatives (Fig. 46a). A series of lab-made modifiers that represent alkynyl derivatives of cyanine dyes (Cy) was described by Gerowska et al.[251] (see Fig. 46b).
Most of studies dealing with the synthesis of oligonucleotide conjugates using click chemistry methods cited in this Section were carried out in homogeneous systems. The CuAAC reaction is conducted under various conditions, in particular using various compositions of the catalytic buffer solution. The most frequently used catalysts are copper(I) halides or copper(II) sulfate in which the metal is preliminarily reduced with ascorbic acid. Meldal and Tornøe[257] noted that a buffer solution containing CuII ions should be preferably used, because the presence of a reducing agent makes it possible to maintain a constant CuI level for binding to alkyne. Also, copper(I) halides require the presence of a base (e.g., amine) in the reaction mixture or elevated temperature.[257] For protecting CuI from oxygen, certain compounds [e.g., tris(benzyltriazolyl)amine (TBTA), N,N,N',N''',N''-pentamethyldiethylenetriamine (PMDETA), or bathophenanthroline disulfonate] are added to the catalytic buffer solution;[257] these additives have a considerable effect on the cycloaddition rate. In contrast to CuAAC, the SPAAC analogue proved to be efficient not only in in vitro, but also in in vivo experiments.[258][259] The reactions involving cyclooctyne derivatives can proceed under physiological conditions and without the use of auxiliary reagents.[260] However, their considerable drawback is the lack of regioselectivity, which results in a mixture of inseparable conjugation products. A number of publications[231][261][262] describe the CuAAC reaction conducted as an automated synthesis using various solvents (1 : 1 ButOH – H2O mixture, DMF, DMSO, acetonitrile, formamide, 1,3-dimethylamylamine (DMAA)] and various copper compounds: salts (CuCl), complexes [Cu(MeCN)4PF6, CuCl · TBTA, CuBr · Me2S, CuBr · PhSMe, CuBr · TBTA, CuI · P(OEt)3], and the CuI – DIPEA system (DIPEA is diisopropylethylamine). It was noted that one problem faced in the use of click reaction is incomplete compatibility with phosphoramidite chemistry, caused by the ability of azides to react with phosphite triesters via the Staudinger reaction pathway (see Section 4.5 below).
4.4. Preparation of conjugates using amino and phosphite or phosphate components
The phosphite [–OP(OCE)O–] (CE is 2-cyanoethyl) and phosphate [(HO)2P(=O)–] moieties of the sugar – phosphate backbone of oligonucleotides can serve as reaction centres for further functionalization, thus providing the preparation of conjugates with various FMs.
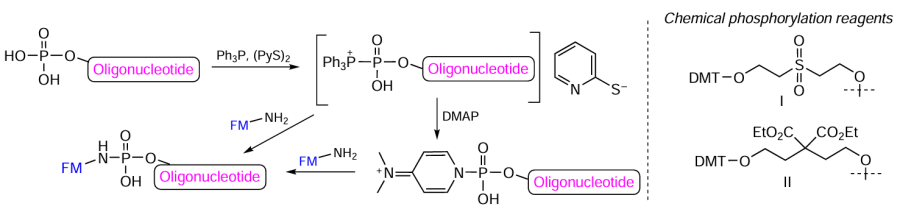
In the early 1970s, T.Mukaiyama and M.Hashimoto[263][264] described in detail the synthesis of nucleotide derivatives and short oligonucleotides using the triphenylphosphine-2,2'-dipyridyl disulfide [Ph3P – (PyS)2] reduction – oxidation coupling reagents. The phosphoroxyphosphonium salts formed upon the reaction of phosphate with this coupling reagents can subsequently react with amines, alcohols, and phosphates to give phosphoramidates, mixed esters of phosphoric acid, and asymmetrical pyrophosphates, respectively.
The proposed coupling reagents by themselves or in combination with a nucleophilic catalyst, 4-dimethylaminopyridine (DMAP) meant for the activation of the terminal phosphate group of fully deprotected oligonucleotide, provide the synthesis of oligonucleotide conjugates with various amino derivatives of biomolecules in a homogeneous process (Fig. 47). For the synthesis of oligonucleotide 5'- and 3'-phosphates, commercial companies offer two types of phosphorylating reagents (I and II, see Fig. 47). The use of these reagents to prepare conjugates with lipophilic, alkylating, and photoactive reactive groups and with polycyclic dyes, antibiotic bleomycin, diamines, and peptides has been described in quite a few publications (e.g.[40][265-270]). Kropacheva et al.[271] proposed a heterogeneous implementation of this method.
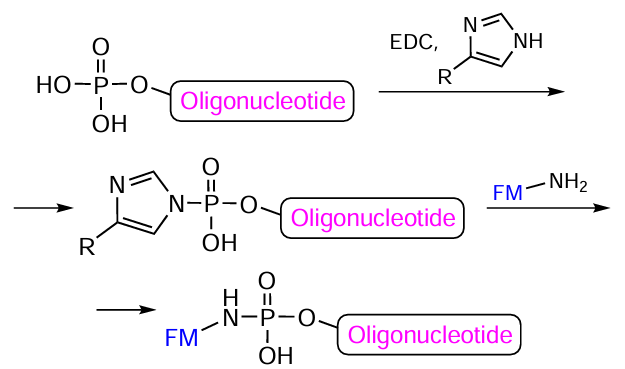
The 5'- or 3'-end of fully deprotected oligonucleotide can also be modified with FMs by activating the terminal phosphoric acid residue with EDC in the presence of imidazole[272] or 4-methylimidazole[273] (Fig. 48).
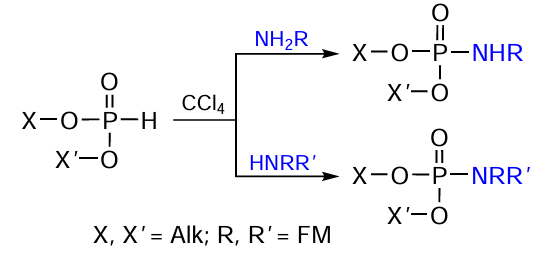
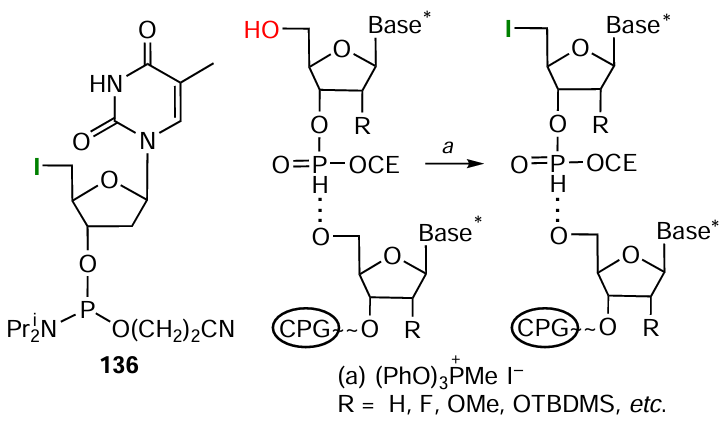
A very convenient and frequently used method for the phosphoramidate bond formation in the heterogeneous synthesis of conjugates proposed in the mid-1940s is based on the Atherton – Todd reaction (АТ). This reaction proceeds between dialkyl phosphite (H-phosphonate) and a primary or secondary amine in the presence of carbon tetrachloride[274] (Fig. 49). The mechanism of this reaction has been studied in considerable detail;[275-278] and it was show that the use of CBr4 , CBrCl3 , or CHI3 instead of CCl4 does not reduce the efficiency of this protocol for the synthesis of phosphoramidates.[275][279-281]
Dialkyl phosphite is formed in the condensation step during the H-phosphonate synthesis of oligonucleotides, with the conditions for its oxidative amination being compatible with the synthetic cycle. The advances in the development of the solid-phase protocol of this synthesis achieved in the late 20th century, together with the simplicity and efficiency of the AT reaction, facilitated the process and made it possible to obtain both short and long mono- and multi-conjugates with various FMs at not only terminal (see Fig. 47, Fig. 48), but also internucleotide phosphate groups.
Currently, nucleoside H-phosphonates are commercially available (Fig. 50), which markedly expands the potential of oligonucleotide conjugate synthesis using the AT reaction with the aim of further studies.
A combination of phosphoramidite and H-phosphonate chemistry (Fig. 51) was proposed by Vlaho et al.[282]The authors incorporated the corresponding nucleoside H-phosphonate into specified positions of a protected immobilized oligonucleotide, performed oxidative amination of the internucleoside phosphite moiety with a number of amines (dimethylethylenediamine, dimethylpropanediamine, dimethylbutanediamine, isopentylamine) in the presence of CCl4, and then continued the chain elongation by the standard automated phosphoramidite method.
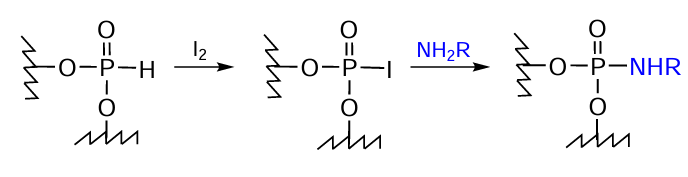
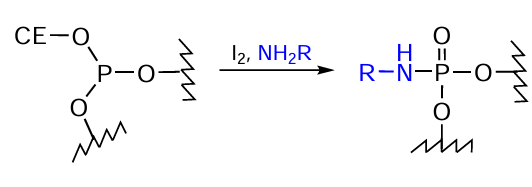
The use of nucleoside H-phosphonates in the solid-phase synthesis makes it possible to introduce various amino-FMs into oligonucleotides via the phosphoramidate bond formation. The heterogeneous protocol alternative to the АТ reaction consists in the oxidation of dialkyl phosphite with elemental iodine[283] followed by replacement of the iodine atom by amino-FMs (Fig. 52).
Mention should also be made of the heterogeneous protocol for the synthesis phosphoramidate oligonucleotide derivatives, which is based on the oxidation of trialkyl phosphite formed in the condensation step during the standard phosphoramidite synthesis with amino-FM in the presence of elemental iodine[284][285] (Fig. 53).
Oligonucleotides modified at the 5'(3')-terminal phosphate groups are mainly obtained in a homogeneous process. The described heterogeneous syntheses of oligonucleotides modified at the internucleotide phosphate linkages cannot, most often, be completely automated.[282][286-289]
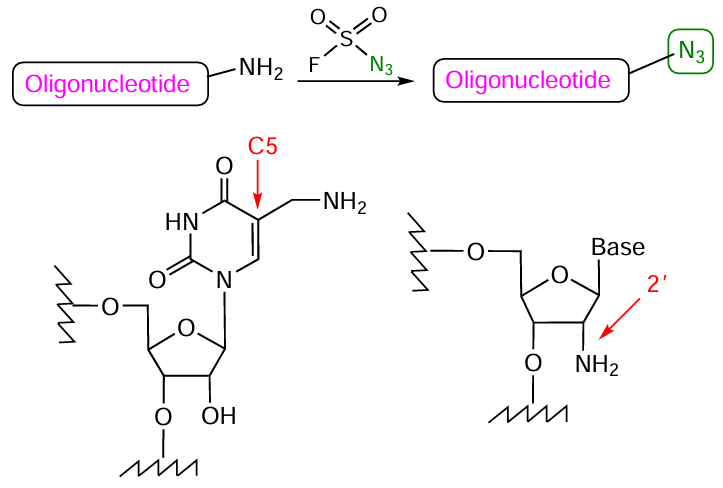
4.5. Preparation of conjugates using azido and phosphite components
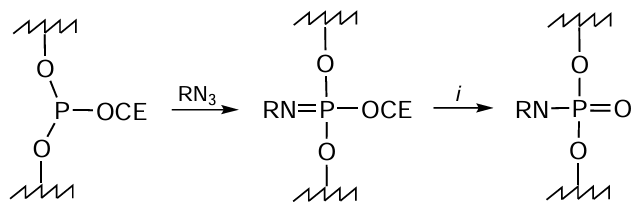
The reaction discovered in 1919 by H.Staudinger[290] and named after him is based on the oxidation of phosphite triesters with organic azides. The Staudinger reaction is widely used for modification of glycans, lipids, DNA, and proteins and for the development of glycopeptides, microchips, and functional biopolymers.[291-293] Now it has become a tool for the preparation of phosphate-modified oligonucleotides and can be used in solid-phase synthesis to functionalize the internucleoside phosphite triester linkage (Fig. 54), which is formed in the coupling step of the standard phosphoramidite synthesis protocol.
As applied to oligonucleotides, this reaction efficiently proceeds if the azido group has an organic substituent with pronounced electron-withdrawing properties (in particular, acyl or sulfonyl substituent). In the case of alkyl azides, elevated temperatures and long reaction times are required;[284][294] this markedly increases the duration of synthesis of the target conjugate, especially when multiple modifications are involved. The azide molecule may also contain additional reactive centres, which allow variation of the substituents.
Biologically active molecules containing carboxyl or aldehyde group can be rather easily converted to acylazido derivatives[197][236] using the following reactions (Fig. 55):*
— direct reaction of carboxylic acid with diphenylphosphoryl azide (1);[295]
— diazotization reaction of sodium azide with activated carboxylic acid derivatives, in particular acid chlorides prepared by treatment of acids with thionyl chloride (2)[296] or formed in situ on treatment of the acid with trichloroacetonitrile and triphenylphosphine (3);[297] benzotriazole acid derivatives obtained by the reaction of acids with 1-(methylsulfonyl)benzotriazole (4);[298] or carboxylic acid hydrazides (obtained by treatment of acids with hydrazine) in the presence of nitrosonium tetrafluoroborate,[299] nitrogen(IV) oxide,[300] or a mixture of nitrogen oxide with oxygen (5);[301]
— one-step conversion of aldehydes on treatment with sodium azide and periodinane (Dess – Martin reaction) (6).[302]
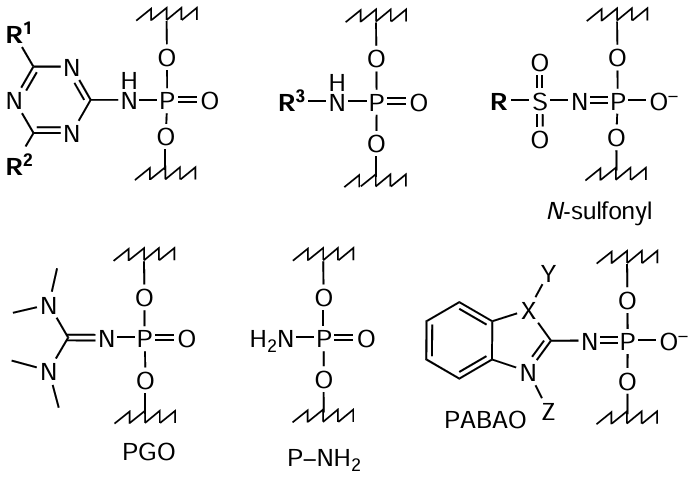
The reaction of azide or acylazide with phosphorus(III) atom is, most often, compatible with the solid-phase synthesis of oligonucleotides and gives a good yield of the product. The active use of this reaction to prepare oligonucleotide derivatives in the last decade not only facilitated the introduction of FM residues at internucleoside phosphate linkages,[294][303-309] but also enabled the synthesis of new charged or electrically neutral phosphate-modified oligonucleotide analogues: N-sulfonyl phosphoramidate, phosphoryl-guanidine (РGO), N-unsubstituted (P – NH2), benzazole (PABAO), and other oligonucleotides possessing altered physicochemical and biological properties compared to those of native oligomers (Fig. 56).[305][309-321]
The introduction of substituents with fundamentally different structural characteristics to the internucleoside phosphate linkages using the Staudinger reaction is a rational and highly efficient approach to the synthesis of a wide range of NA conjugates and analogues containing P – N bonds. This has substantially expanded the potential for modification and functionalization of oligonucleotides.
* The preparation of azido-FMs is described in Section 4.3.
4.6. Preparation of conjugates using carbonyl and amino or hydrazido components
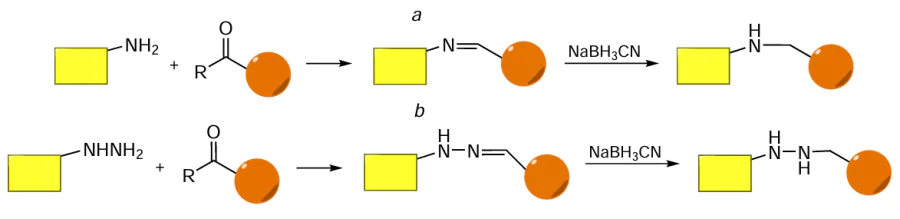
Aldehydes and ketones containing a formyl and acyl group, respectively, can react with amines to give azomethines, Schiff bases,[322][323] named after Italian chemist who discovered this reaction in 1864 (Fig. 57a). A similar reaction with hydrazine derivatives (see Fig. 57b) yields the corresponding hydrazones. The azomethine moiety (–N=C–) is acid-labile; its sensitivity to pH of the medium depends on the structure of adjacent groups.[324][325] For stabilization of conjugates of this type, the azomethine moiety is often reduced to the stable C – N bond by treatment with, for example, sodium cyanoborohydride (see Fig. 57).
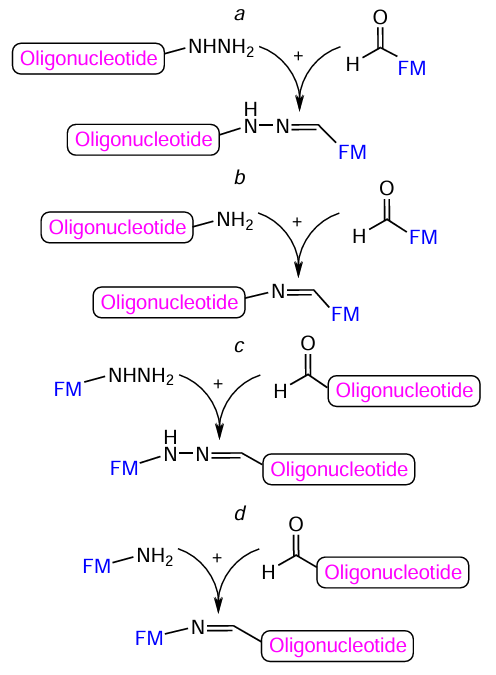
Oligonucleotide — FM conjugates are prepared using coupling of an amine (or hydrazine) component only with a formyl group, according to the four reaction pathways depicted in Fig. 58.
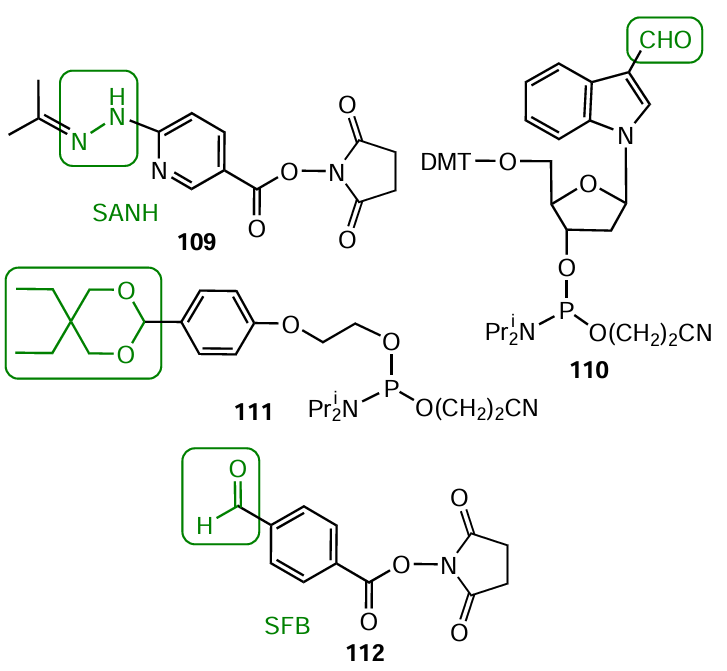
Commercial reagents are available for all four coupling pathways. The preparation of amino-containing oligonucleotides using modifiers was described above (see Section 4.1). The introduction of a hydrazido group into oligonucleotide can be performed indirectly, for example, by treating an amino-modified oligomer with bifunctional 2,5-dioxopyrrolidin-1-yl-6-[2-(propan-2-ylidene)hydrazinyl]nicotinate (SANH) 109 (Fig. 59). Two types of modifiers have been proposed for the introduction of aldehyde group to the 5'-end of the oligonucleotide chain. The first one is nucleoside-like phosphoramidite 110 with an exocyclic aldehyde group in the indole ring; it can be used in any cycle (intermediate or final) of the automated oligonucleotide synthesis. The second one is terminal non-nucleotide modifier 111 containing a protected aldehyde group and meant for the final cycle of the synthesis of pre-activated derivative. Alternatively, an aldehyde group can be indirectly introduced into oligonucleotide via the corresponding amino derivative (see Section 4.1), using, for example, bifunctional reagent 112: 2,5-dioxopyrrolidin-1-yl 4-formylbenzoate (SFB).
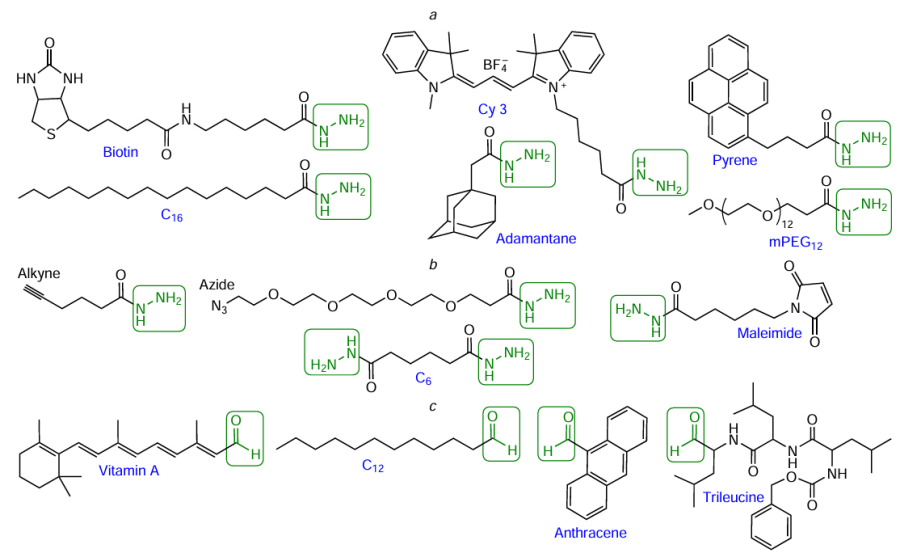
In view of the high efficiency of reactions involving aldehyde and hydrazide groups, manufacturers produce a wide range of commercially available hydrazide-containing dyes, quenchers, and some other FMs. This is exemplified in Fig. 60a, which shows hydrazide derivatives of biotin, cyanine dye Cу3, polyethylene glycol (mPEG12), and adamantaneacetic, pyrenebutyric, and hexadecanoic (C16) acids. Special mention should be made of hetero- or bifunctional hydrazide-containing reagents (see Fig. 60b): the hydrazide component actively reacts with aldehydes; compounds containing alkynyl or azido groups are used in CuAAC or SPAAC reactions (see Section 4.3); the maleimide moiety is involved in thiol – maleimide reaction (see Section 4.7 below). Meanwhile, commercially available FMs containing an aldehyde group are not as widely available. As examples of these compounds, note anthracene-9-carbaldehyde, retinal (vitamin A aldehyde), lauraldehyde, and the formyl derivative of the peptide trileucine (see Fig. 60с).
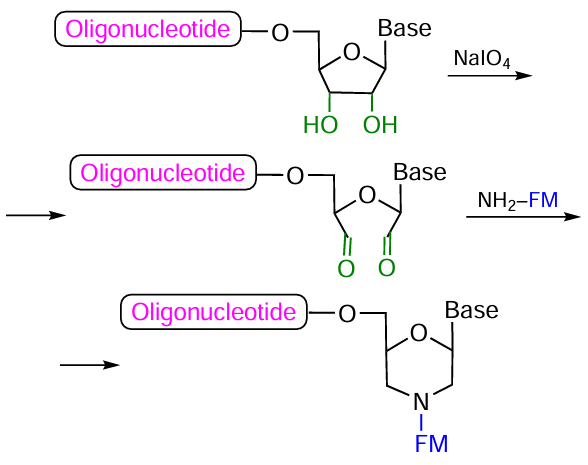
As regards conjugation using lab-made modifiers, note a facile and efficient method for the generation of aldehyde groups at the oligonucleotide 3'-end consisting in the oxidation of the 3'-terminal ribose residue with sodium periodate[326-328] in solution (Fig. 61). The reaction of the resulting 2',3'-diformyl oligomer with a primary aliphatic amine within FM affords a conjugate by means of formation of a substituted morpholine ring.
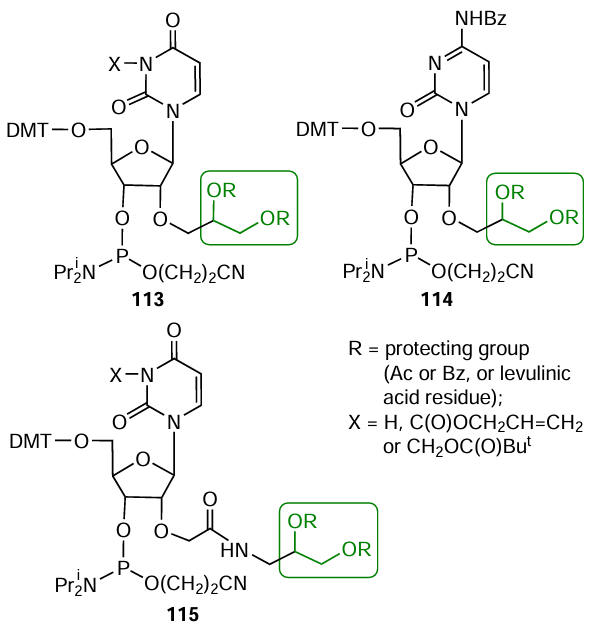
As further development of this strategy, it was proposed[174][329-334] to prepare nucleotide modifiers 113 – 115 containing initially protected diol groups in 2'-position of ribose; subsequently, these groups can generate a 2'-formyl group upon the reaction with NaIO4 (Fig. 62). Most often, benzoic, levulinic, and acetic acid residues were used to protect the OH groups in diols.
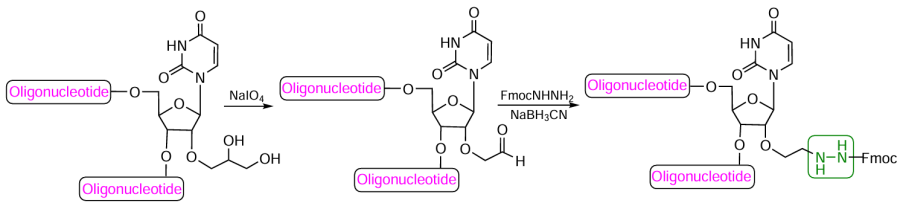
Phosphoramidites 113 – 115 can also be used for indirect introduction of a hydrazido group in any position of the oligonucleotide chain.[330][331][334-338] Thus after treatment of fully deprotected oligonucleotide with NaIO4 and conversion of the 1,2-diol group to an aldehyde group, a Fmoc-protected hydrazide was introduced in this position, and then the resulting azomethine bond was reduced with sodium cyanoborohydride (Fig. 63). The subsequent removal of the Fmoc protection under mild conditions furnished the desired 2'-hydrazide-containing oligonucleotides.
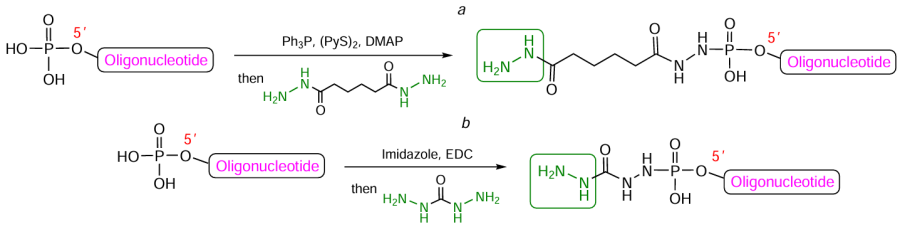
Hydrazide-containing compounds can react not only with aldehyde oligonucleotide derivatives, but also with oligomers carrying, for example, 5'-phosphorylated chains (similarly to the approach described in Section 4.4). For example, the use of the Ph3P – (PyS)2 reduction – oxidation coupling reagents in combination with DMAP as a nucleophilic catalyst allows for highly efficient attachment of the commercially available adipic acid (C6) dihydrazide residue (see Fig. 60b) to the oligonucleotide end (Fig. 64a).[339] Ghosh et al.[340] activated the terminal phosphate group toward oligomer 5'-modification with carbohydrazide using EDC in the presence of imidazole (see Fig. 64b).
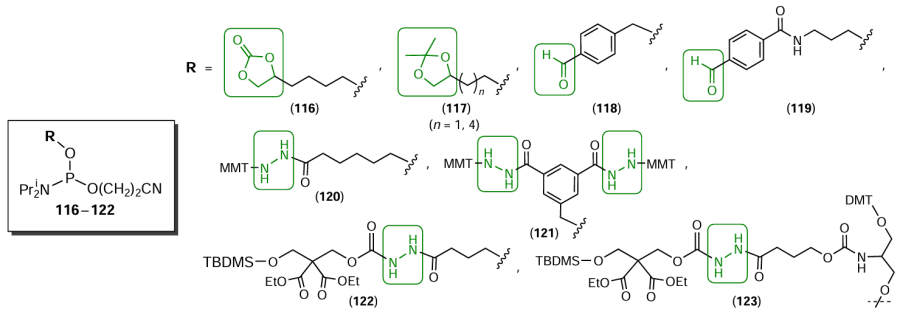
Typical lab-made terminal modifiers containing aldehyde (compounds 116 – 119) and hydrazide (compounds 120 – 123) functional groups, or their precursors, are shown in Fig. 65 and are described in quite a number of studies.[341-345] It is worth noting that compound 123 acts as a modifier that allows a hydrazido group to be introduced in any position in the newly formed oligonucleotide chain.[343]
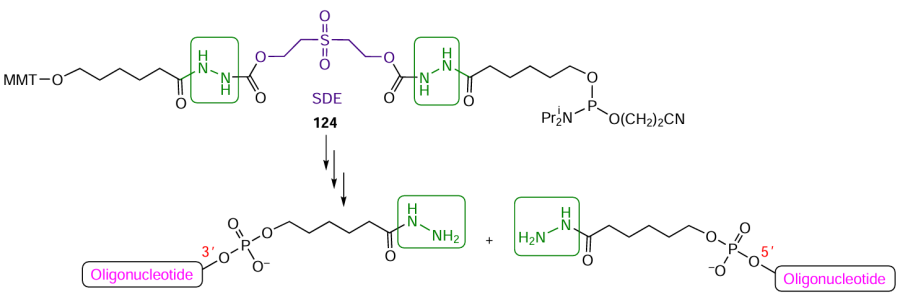
An original method for the preparation of two oligonucleotides modified with 3'- or 5'-hydrazide groups in one automated synthesis has been proposed.[343][346] Using ε-caprolactone, hydrazine, and sulfonyl diethanol moieties, the authors synthesized non-nucleoside phosphoramidite modifier 124 (Fig. 66) able to generate terminal hydrazide groups upon elimination of the sulfonyl diethanol (SDE) moiety via the final ammonolysis deprotection (by analogy with Refs [347] and [348]). This results in the formation of two oligonucleotides, each containing a hydrazido group attached to the phosphate group at the 5'- and 3' ends of the oligomeric chain.
The preparation of amino-FMs that are not commercially available was described above (see Fig. 30). Among the laboratory procedures for the introduction of formyl groups into FMs that are summarized in Fig. 67, note the following reactions:
— selective catalytic reduction of acyl chlorides with hydrogen to aldehydes,[349][350] which is usually catalyzed by lithium tri(tert-butoxy)aluminium hydride or by palladium metal supported on various materials (BaSO4, CaCO3, ВaCO3, asbestos, coal, diatomaceous earth) (1);
— reduction of esters to aldehydes using diisobutylaluminium hydride at low temperatures (2);[351]
— oxidation of aliphatic hydroxyl group with copper(II) oxide, manganese(IV) oxide, silver carbonate on zeolite, chromium(VI) oxide complexes with tertiary amines (Sarett – Collins reagent, CrO3 · 2 C5H5N),[352][353] Corey reagent (pyridinium chlorocromate, [C5H5NH]+[CrO3Cl]–)[354] (3), and oxalyl chloride with dimethyl sulfoxide in the presence of a base (Svern oxidation)[355][356] (4);
— oxidation in the presence of nitroxide: 2,2,6,6-tetramethylpiperidin-1-oxyl (5).[357]
The most widely used method for the synthesis of FM hydrazides is the acylation of hydrazine with activated esters, anhydrides, or carboxylic acid chlorides[358][359] (Fig. 68).
Thus, despite the wide range of both commercial and lab-made modifiers available, this reaction is implemented, if necessary, using amino-containing oligonucleotides (see Section 4.1) in combination with SANH (109) and SFB (112) bifunctional reagents (see Fig. 59). This approach provides the synthesis of oligonucleotides modified at any position with a formyl or hydrazido group.
4.7. Preparation of conjugates using thiol, maleimido, and halido components
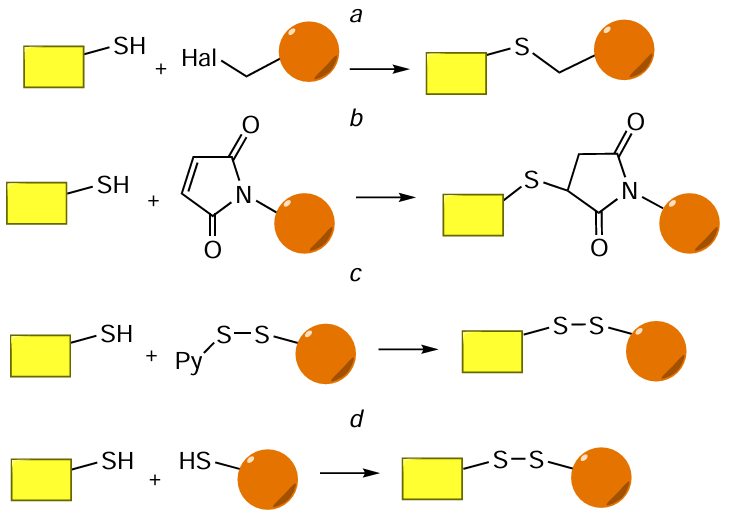
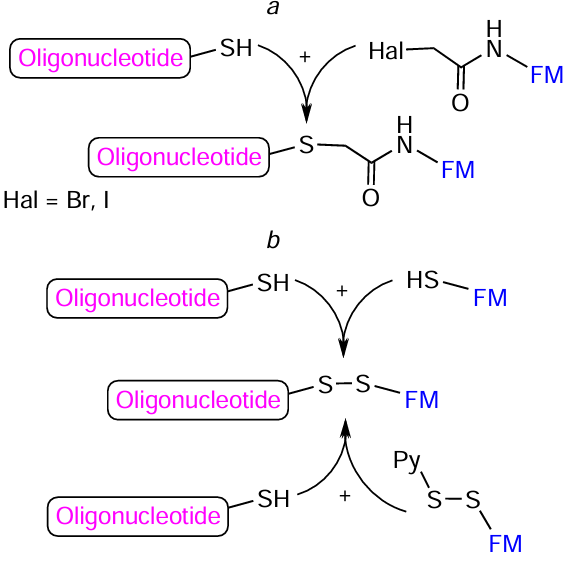
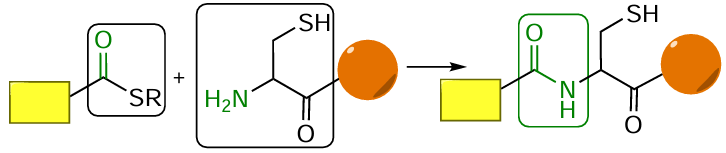
Another type of reactions widely used to produce oligonucleotide conjugates are reactions involving thiol (SH) groups: alkylation of thiols with alkyl halides, most often iodides (Fig. 69a), thiol – maleimide (thiol – ene) reaction (see Fig. 69b), thiol – disulfide (thiol) exchange (see Fig. 69c), and sulfhydryl oxidative addition (see Fig. 69d ). The alkylation of thiols or thiolates with alkyl halides is the Williamson reaction adapted to thiols;[360] this reaction named after the English chemist was discovered in 1850 for the preparation of unsymmetrical ethers. The thiol – ene reaction has been known for more than 120 years, since T.Posner[361] carried out the reaction of mercaptans with unsaturated hydrocarbons back in the early 1900s. The method of controlled S – S bond formation based on the thiol – disulfide exchange was proposed in 1970 by V.P.Saxena and D.Wetlaufer.[362] The role of thiols, which can be oxidized by a wide variety of oxidants to give disulfides, in metabolic processes in living systems was discovered in the late 1940s and early 1950s, when a group headed by German biochemist F.Lipmann[363][364] showed that the activity of coenzyme A is due to the presence of the mercaptoethylamine SH group.
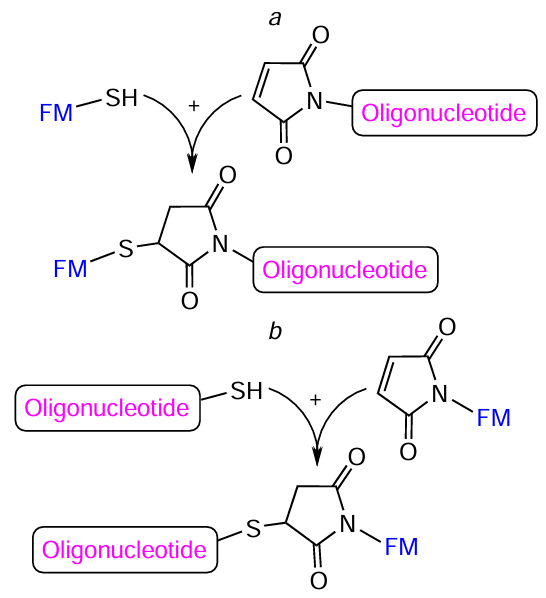
In fact, the thiol – maleimide reaction giving a stable thioether bond has become the most common method for the synthesis of oligonucleotide – FM conjugates, irrespective of the arrangement of reactive groups in the components (Fig. 70).
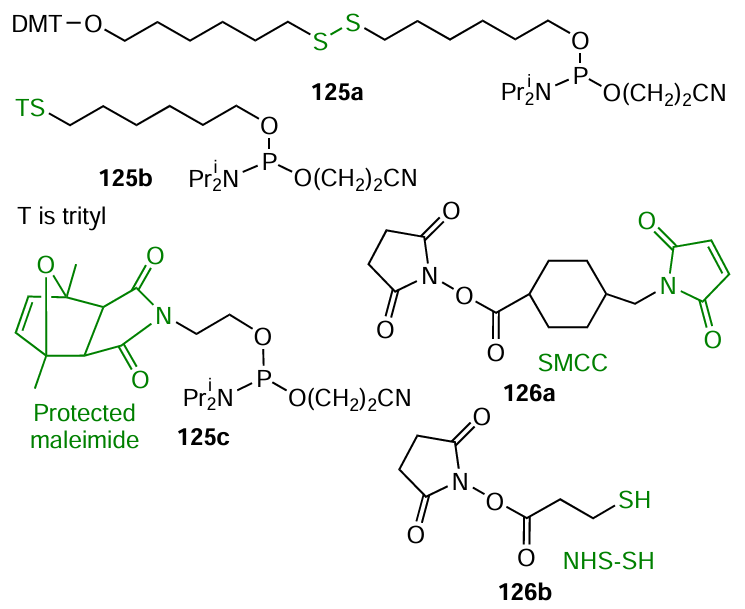
Commercial modifiers for oligonucleotides containing protected thiol groups are widely used in automated oligonucleotide synthesis (Fig. 71). A free thiol group is formed, for example, when compounds 125a,b are treated with dithiothreitol or silver(I) nitrate, respectively. A possible alternative approach to thiol-dependent conjugation is the use of the 5'-terminal phosphoramidite modifier 125c with protected maleimide moiety, which is regenerated via the [4 + 2]-cycloelimination (retro-Diels – Alder reaction).[365] A convenient method for additional functionalization of amino-containing oligonucleotides (see Section 4.1) is the use of bifunctional linker-forming molecules 126a,b: N-hydroxysuccinimide esters of 4-(N-maleimidomethyl) cyclohexanecarboxylic (SMCC) and 3-mercaptoropionic (NHS – SH) acids.
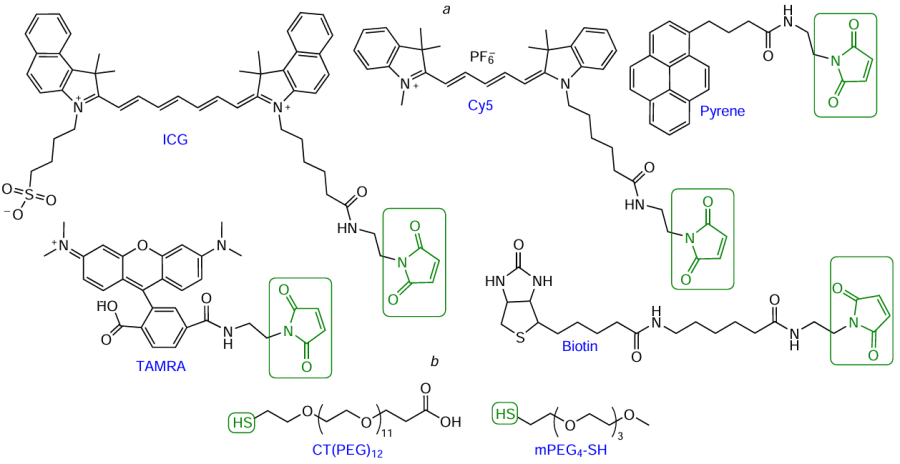
The range of commercially available maleimide modifiers is quite extensive. As an example, Fig. 72a shows maleimides based on indocyanine green (ICG), cyanine (Cу5), and xanthene (TAMRA) dyes, pyrenebutyric acid, and biotin. Commercially available thiol reagents (see Fig. 72b) mainly include various SH-substituted polyethylene glycols [e.g., CT(PEG)12, mPEG4-SH].
The Williamson reaction (initially developed for alcohols) was adjusted for thiol-containing oligonucleotides and iodo- and bromoacetamide FMs (Fig. 73a). In particular, it was shown that in the case of peptides, conjugation can be carried out over a few hours at room temperature (e.g.[366-369]). The oxidation of sulfhydryl groups, like the thiol – disulfide exchange (see Fig. 73b), leads to the formation of conjugates in which FM and oligonucleotide are linked by biolabile S – S bond (e.g.[370-372]).
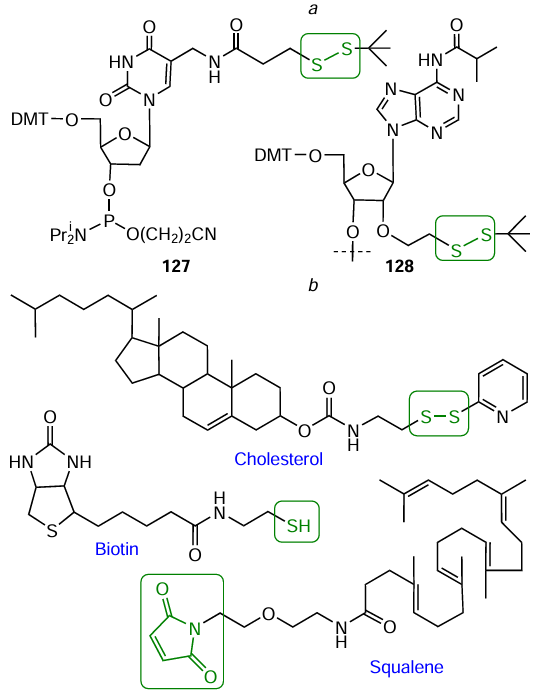
A number of lab-made nucleotide SH-containing modifiers convenient for the synthesis of oligonucleotide conjugates with various FMs have been reported in the literature. Kupihár et al.[373] introduced S – S-containing group in thymidine C(5) position using 3-(2-tert-butyldisulfanyl)-thiopropionic acid to give modifier 127 (Fig. 74a). Solid support 128 containing 2'-O-(2-tert-butyldisulfanylethyl)-N 6-isobutyryladenosine was prepared via a nine-step synthesis from 2'-O-bromoethyl adenosine dertivative.[374] Examples of lab-made non-nucleotide FMs containing free and protected thiol group or maleimide moiety[372][375][376] are depicted in Fig. 74b.
In this Section, particular attention should also be paid to so-called native chemical ligation, one of the effective methods for amide bond formation upon conjugation of thiocarboxylic acid derivatives and cysteine-containing molecules. Historically, this approach was proposed for the synthesis of long peptides, since this makes it possible to extend the length of the peptide chain during chemical synthesis by more than 50 amino acid residues. Chemoselective native ligation reaction was proposed[377] in 1994 for the reaction between unprotected peptides, one containing C-terminal thioether and the other containing N-terminal cysteine (Fig. 75). It is noteworthy that this process occurs not only in an aqueous solution, but also in the presence of denaturing agents and organic solvents and, hence, it enables cross-linking of both hydrophilic and hydrophobic moieties of biologically active molecules. This type of conjugation is most appropriate for the synthesis of oligonucleotide conjugates with peptides or other FMs containing a cysteine residue (e.g.[378-384]).
As can be seen from the above, conjugation involving SH groups is quite popular for modification of biomolecules, particularly proteins, peptides, and antibodies, which typically contain thiol groups.[201] [385-393] The Williamson reaction is mainly used in the synthesis of peptide conjugates. A key benefit of the maleimide – thiol reaction is the mild reaction conditions, which ensure that complex biomolecular structures remain virtually intact. The formation or cleavage of the S – S disulfide bond is a reversible redox reaction determining particular applications of the corresponding conjugates.
4.8. Preparation of conjugates using halido and amino or alkynyl components
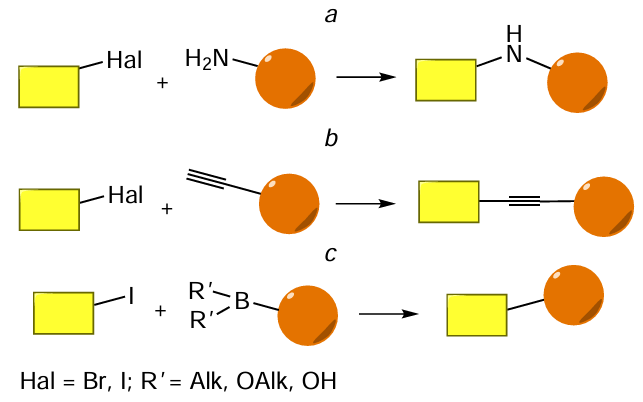
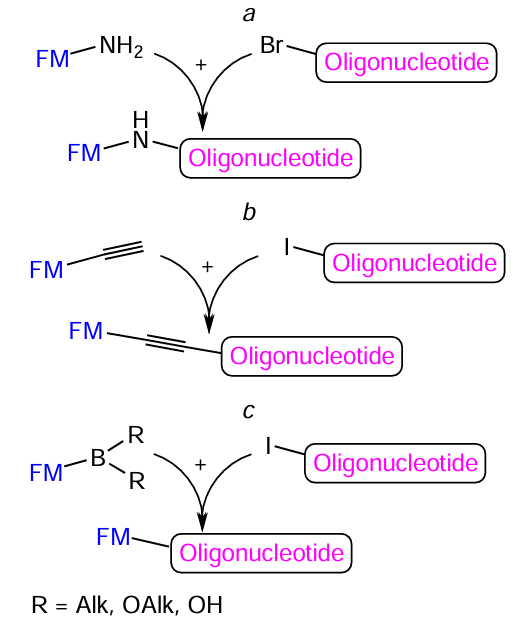
Molecules containing an amino group are able to react with haloalkanes by substituting the halogen atom according to the N-alkylation pathway (Fig. 76a). Another type of conjugation involving a halogen atom is the palladium- and copper-catalyzed formation of a carbon – carbon bond (see Fig. 76b,c). The alkynylation of aryl halides using aromatic acetylenes was first described in 1975 by three independent research groups: L.Cassar,[394] H.A.Dieck and R.F.Heck,[395] and K.Sonogashira et al.[396] All reactions proceed in the presence of palladium catalysts; however, the first two protocols require harsh conditions (such as high temperatures). The use of copper co-catalyst, apart from palladium complexes, in the K.Sonogashira method[396] made it possible to conduct the reactions under mild conditions to obtain products in good yields. In the Suzuki (or Suzuki – Miyaura) reaction first described in 1979, N.Miyaura and A.Suzuki[397] (see Fig. 76с) used a complex palladium-based catalyst for the reaction of arylboronic acids with halogenated organic molecules. The Suzuki and Sonogshira reactions represent cross-coupling reactions and can occur selectively for reactants containing diverse functional groups, which is especially important for the synthesis of complex molecules.
The oligonucleotide conjugates with various FMs are synthesized, most often, by reactions of halogen-containing oligonucleotides (halogen is bromine or iodine) with amino-, alkynyl-, or boron-modified FMs (Fig. 77). As a rule, the reactions are conducted as heterogeneous processes.
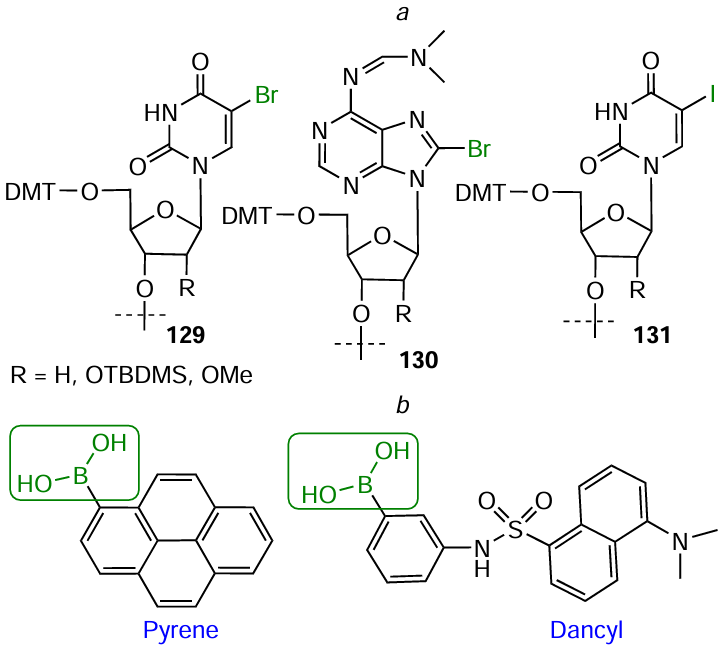
The conjugation according to the above reactions can be performed using commercial nucleotide phosphoramidites or solid supports 129 – 131 containing a halogen atom in a heterocyclic base, adenosine or uridine (Fig. 78a). As examples of commercially available boron-containing FMs, consider pyren-1-yl- and 3-(dansylamino)phenylboronic acids (Fig. 78b). The amino- and alkynyl-FMs are described in Sections 4.1 and 4.3, respectively.
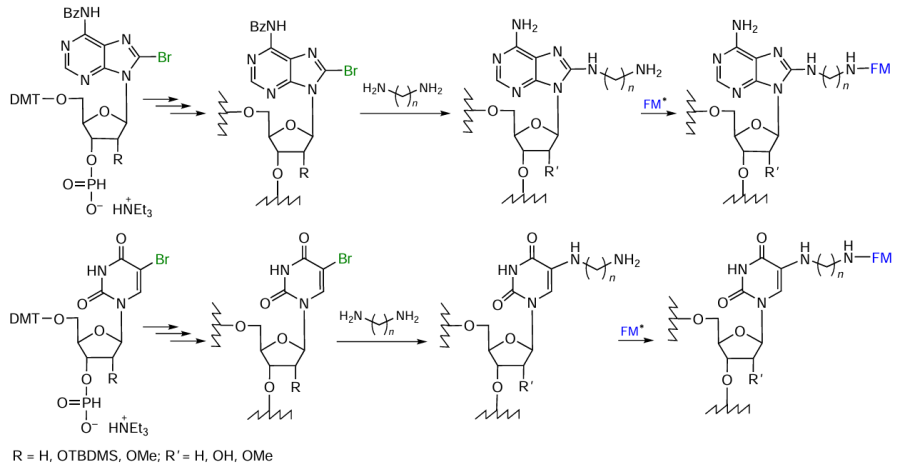
Repkova et al.[398-400] using the H-phosphonate method of oligonucleotide synthesis and lab-made H-phosphonates of 5-bromouridine and 8-bromoadenosine carried out the introduction of diamines into heterocyclic bases (Fig. 79). The amino-modified oligonucleotides were further used to prepare oligonucleotide derivatives containing nitroxide labels or perfluoroaryl azide groups. These oligomers can serve, for example, for studying the structural and functional topography of human ribosomes.[401-407]
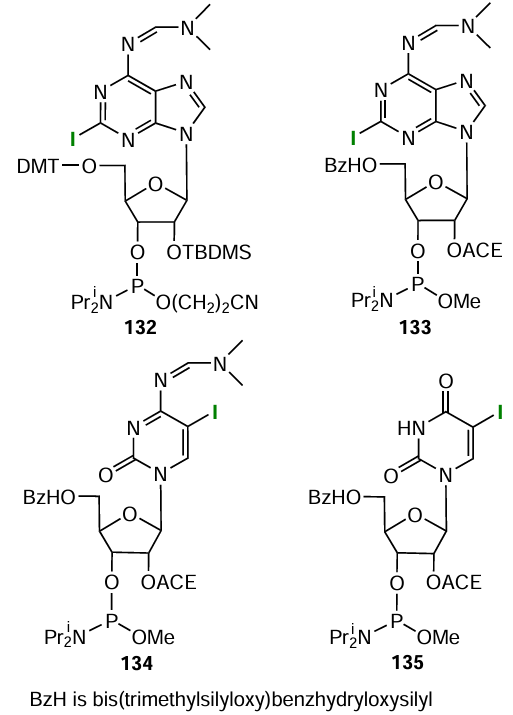
Piton et al.[408] prepared nitroxide-labelled conjugates by the Sonogashira reaction with iodinated nucleotide modifiers: commercial 5'-O-dimethoxytrityl-2'-O-tert-butyldimethylsilylnucleoside-phosphoramidite 132 (Fig. 80), meant for standard phosphoramidite synthesis, and lab-made modifiers 133 – 135. The latter represented ribophosphoramidites containing 2'-bis(acetoxyethoxy)methyl (АCЕ) protecting group, which is suitable for automated synthesis according to the 2'-ACE protocol proposed by Dharmacon™ (see Fig. 80).
The replacement of the 5'-terminal hydroxyl group in oligonucleotide by an iodine atom also enables successful conjugation with FMs in a heterogeneous process. The 5'-iodo derivatives can be prepared either using a commercial modifier, 5'-terminal phosphoramidite 136 already containing a iodine atom (Fig. 81), or by converting oligonucleotide resulting from automated synthesis to 5'-iodo derivative[409] according to the scheme given in the Figure.
The iodine atom at the oligonucleotide 5'-end can also be replaced by an azido, amino, or thiol group in high yield, as described in the literature[410-413] (Fig. 82). The resulting 5'-amino derivative can react with NHS esters of FMs (see Fig. 20, Fig. 23) or with formyl-containing FMs (see Fig. 59, Fig. 60, Fig. 67). The azido group at the oligonucleotide 5'-end is suitable for the azide – alkyne cycloaddition (see Fig. 41), while 5'-SH derivatives can be converted to the corresponding conjugates by the reactions presented in Fig. 70 and Fig. 73.
The reactions described in this Section are widely used both for modification of oligonucleotides as a whole and for the preparation of various types of modifiers (e.g.[414-420]).
Considering the diversity of reactive groups discussed in this review capable of specific (orthogonal) interaction with one another, the potential applications of the post-synthetic approach are virtually unlimited and make it possible to obtain not only mono- but also multifunctionalized conjugates.
5. Conclusion
Oligonucleotides and their derivatives and conjugates play a key role in molecular biology, biomedicine, and pharmaceutics, where they act as molecular probes and therapeutic agents for genetic, infectious, and other diseases. The presented review considers methods and approaches that allow rational (i.e., consisting of the minimum number of chemical reactions) synthesis of the target oligonucleotide conjugates with FMs using effective reagents. Rational planning of the synthesis protocol for the desired oligonucleotide construct largely determines the synthesis efficiency, as evidenced by high yields of the target products. In addition, after this modification, it is important to preserve the functional properties of the biologically active compound and the NA component, which is specifically designed for interaction with a particular biological target. The developed protocols are implemented using both commercially available and specially laboratory-made modifiers meant for addressing particular experimental requirements according to two approaches. The pre-synthetic approach implies the use of ready-made (already containing FM) synthons, nucleotide and non-nucleotide ones. This is convenient and justified, but is limited to a specific set of functions. The post-synthetic approach is based on the introduction of reactive groups into both components, which allows for various conjugation options between NA and FM, while also taking into account their chemical stability, reactivity, and the conditions of the relevant reactions.
The data presented in this review make it possible to develop strategies and to plan synthesis options for oligonucleotide derivatives and functional residues and to consider process simplicity and facility. In addition, it is noteworthy that a variety of products can be fabricated on the basis of one oligonucleotide construct. Thus, the review defines the principle of the ‘oligonucleotide construction set’, manipulation of components of which would give rise to a rational protocol for the synthesis of a desired oligonucleotide — FM conjugate possessing specified properties and suitable for successful use for research or practical purposes. The review provides compelling evidence that a wide variety of approaches to the design of nucleic acid conjugates are currently available. This makes it possible to fabricate oligonucleotide conjugates with relatively simple small molecules and with complex macromolecular functional compounds with virtually no limitations. The conjugation strategies presented here offer the opportunity to select a promising synthetic route for complex molecules or to highlight the need to develop new synthetic approaches to oligonucleotide conjugates with biomolecules possessing a predicted structure and unknown but potentially promising function.
This review was written within the State Assignment of the Institute of Chemical Biology and Fundamental Medicine, Siberian Branch, Russian Academy of Sciences, No. 125012300656-5; Sections 4.4 and 4.5 were supported by the Russian Science Foundation (Project No. 21-64-00017П).
6. List of abbreviations and symbols
The following abbreviations and symbols are used in the review:
АТ — Atherton Todd (reaction),
АCЕ — 2'-bis(acetoxyethoxy)methyl,
ABPD — 2-(4-aminobutyl)propane-1,3-diol,
Base — heterocyclic base,
CDI — carbonyldiimidazole,
CЕ — 2-cyanoethyl,
CPG — controlled pore glass or another suitable solid support,
CuAAC — Cu-catalyzed azide – alkyne cycloaddition,
DBCO-amine — dibenzocyclooctyne-amine,
DCC — N,N'-dicyclohexylcarbodiimide,
DEA — diethanolamine,
DHA — docosahexaenoic acid,
DIC — N,N'-diisopropylcarbodiimide,
DIPEA — diisopropylethylamine;
DMAA — 1,3-dimethylamylamine,
DMAP — 4-dimethylaminopyridine,
DMT — 4,4'-dimethoxytrityl,
DMS(O)MT — 4,4'-dimethoxy-4''-sulfonyltrityl,
DSC — N,N-disuccinimidyl carbonate,
EPA — eicosapentaenoic acid,
EDC — 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide,
EtSTetr — 5-ethylthio-1H-tetrazole,
FM — functional molecule,
Fm — 9-fluorenylmethyl,
Fmoc — 9-fluorenylmethoxycarbonyl,
GalNAc — N-acetylgalactosamine,
HHPyr — 4-hydroxy-2-(hydroxymethyl)pyrrolidine,
HATU — hexafluorophosphate 2-(1H-7-azabenzotriazol-1-yl)-1,1,3,3-tetramethyluronium,
HBTU — hexafluorophosphate 2-(1H-benzotriazol-1-yl)-1,1,3,3-tetramethyluronium,
HOAt — 1-hydroxy-7-azabenzotriazole,
HOBt — 1-hydroxybenzotriazole,
MMТ — monomethoxytrityl,
Ms — methanesulfonyl,
Myr — myristic acid,
NA — nucleic acid,
NHS — N-hydroxysuccinimide,
PCho — phosphocholine,
PEG — polyethylene glycol,
PFP — pentafluorophenol,
PMDETA — N,N,N',N'',N''-pentamethyldiethylenetriamine,
PNA — peptidyl nucleic acid,
Py — 2-pyridine,
SANH — 2,5-dioxopyrrolidin-1-yl-6-[2-(propan-2-ylidene)hydrazinyl]nicotinate,
SDE — sulfonyldiethanol,
SFB — 2,5-dioxopyrrolidin-1-yl 4-formylbenzoate,
SPAAC — strain-promoted azide-alkyne cycloaddition,
TBDMS — tert-butyldimethylsilyl,
TBTA — tris(benzyltriazolyl)amine,
ТC — 1,1-dioxo-1λ(6)-thiomorpholine-4-carbothioate (thiocarbamate group),
ТOM — 2'-O-triisopropylsilyloxymethyl,
Tris — tris(hydroxymethyl)aminomethane.
References
































































































































































![Simple and facile reduction of azides to amines: synthesis of DNA interactive pyrrolo[2,1- c ][1,4]benzodiazepines](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)

























![Peptidotriazoles on Solid Phase: [1,2,3]-Triazoles by Regiospecific Copper(I)-Catalyzed 1,3-Dipolar Cycloadditions of Terminal Alkynes to Azides](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)



































![A Strain-Promoted [3 + 2] Azide−Alkyne Cycloaddition for Covalent Modification of Biomolecules in Living Systems](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)



































































![Oligonucleotides containing 2′-O-[2-(2,3-dihydroxypropyl)amino-2-oxoethyl]uridine as suitable precursors of 2′-aldehyde oligonucleotides for chemoselective ligation](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)