Keywords
Abstract
The development of effective encapsulation methods for hydrophobic compounds is a relevant interdisciplinary challenge, particularly for medicine and biotechnology where high therapeutic potential of compounds often cannot be implemented due to solubility and delivery issues. This review provides a systematic analysis of one-step ultrasonic synthesis of emulsion capsules composed of polymers. According to this approach, ultrasonic cavitation not only facilitates the formation of stable emulsions, but also initiates a cascade of physicochemical transformations of biopolymers (proteins, polysaccharides, and glycoproteins), resulting in the formation of robust shells at an interface. This review presents the first comparison and classification of shell formation mechanisms depending on the polymer type, clarifies the role of generated radicals, and highlights the synergistic effects observed in hybrid systems. Special emphasis is placed on the practical potential of this technique for the design of drug delivery systems, contrast agents, and smart materials with controlled release functionality.
The bibliography includes 93 references.
1. Introduction
Currently, studies on the development of new carriers for hydrophobic functional compounds are being actively carried out all over the world. The use of these compounds is highly limited because of difficulties associated with their distribution in aqueous media, in particular the fact that they cannot spontaneously move in the bloodstream. Therefore, delivery of hydrophobic compounds is a highly relevant issue in biomedicine. This issue is addressed using conjugates, micelles, particles, and capsules of various types capable of encapsulating and transporting hydrophobic molecules in aqueous media. Meanwhile, capsules with an oil core offer a number of advantages over other delivery systems: high loading capacity, the presence of the target substance in liquid or dissolved state, and extensive possibilities for designing systems with high mechanical and aggregative stability using various types of biocompatible and biodegradable materials.
Capsules with hydrophobic content are mainly produced by numerous emulsification methods that have been long used in the production of food products, cosmetic products, fertilizers, and so on. However, emulsions are thermodynamically unstable; therefore, for practical applications, it is often necessary to prepare emulsion capsules: systems based on emulsions that do not break down when the internal or external phase is removed or replaced. There are several approaches to the fabrication of emulsion capsules, including stabilization of the emulsion droplets by amphiphilic copolymers[1] and by micro- or nanoparticles (Pickering emulsions),[2-4] in situ polymerization, and interfacial deposition induced by solvent evaporation or replacement.[2] The formation of capsules induced by low-frequency ultrasound is an actively developing method.[5][6] The benefits and drawbacks of the most popular methods used to manufacture emulsion capsules are compared in Table 1. The benefits of the ultrasonic method open up extensive prospects for the use of ultrasound for encapsulation of quite a few hydrophobic compounds in various fields of science and engineering, including biotechnologies, medicine, pharmacy, and agriculture.
The main driving force of the processes that occur in solutions under the action of ultrasonic waves is ultrasonic cavitation. The extreme conditions resulting from the collapse of cavitation bubbles cause the dissociation of water molecules, leading to the formation of hydroxyl and hydrogen free radicals,[7] which act as primary oxidants and reducing agents and can also initiate secondary chemical reactions.[8-10] One of the reactions yields superoxide, which actively initiates chemical transformations of molecules. In the case of synthesis of emulsion capsules using low-frequency ultrasound, cavitation bubbles collapse most often at the oil/water interface, resulting in the formation of a polymer shell on the surface of the dispersed phase droplets, which ensures the mechanical strength of the capsules.
Despite the fact that the ability of ultrasound to induce chemical reactions in polymers has been known since the 1930s, the main focus of research has been limited to polymerization processes and the synthesis of block copolymers. The ultrasonic synthesis of emulsion capsules was first described in 1990, when aqueous suspensions of protein microcapsules loaded with hydrophobic liquids were prepared for the first time.[5] Subsequently, this process was used to form capsules of polyglutamic acid, avidin, and streptavidin.[11-13] Later, an approach was proposed to obtain a-amylase microspheres using ultrasound; in the microspheres formed in this way, the enzyme retained approximately 56% of its specific activity.[14] Currently, ultrasonic synthesis is being rapidly developed and serves both for the production of capsules with single-component shells made of natural polymers, such as nucleic acids, for the delivery of genetic material[15] and for the fabrication of capsules based on polyelectrolyte complexes made of two oppositely charged polysaccharides.[16]
In this paper, we consider characteristic features of the production of biopolymer capsules using low-frequency ultrasound, describe the available publications on the synthesis of this type of capsules, give classification of the used macromolecules, and consider in detail the mechanism of formation of polymer shells depending on the set of the chosen components. The understanding of the mechanism of formation of capsule shells, in particular the role of ultrasonication in this process, lays the groundwork for further expansion of this approach to the development of new core – shell materials, which hold promise as drug delivery systems and components of new-generation coatings.
2. The role of ultrasonic emulsification in the formation of the emulsion polymer capsules
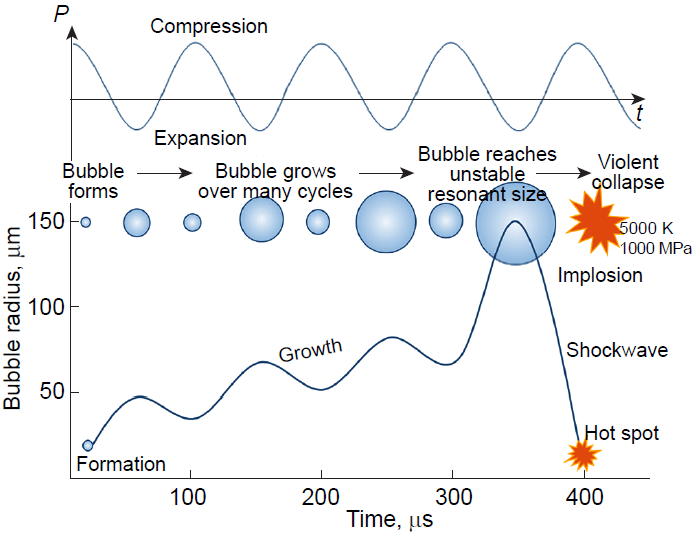
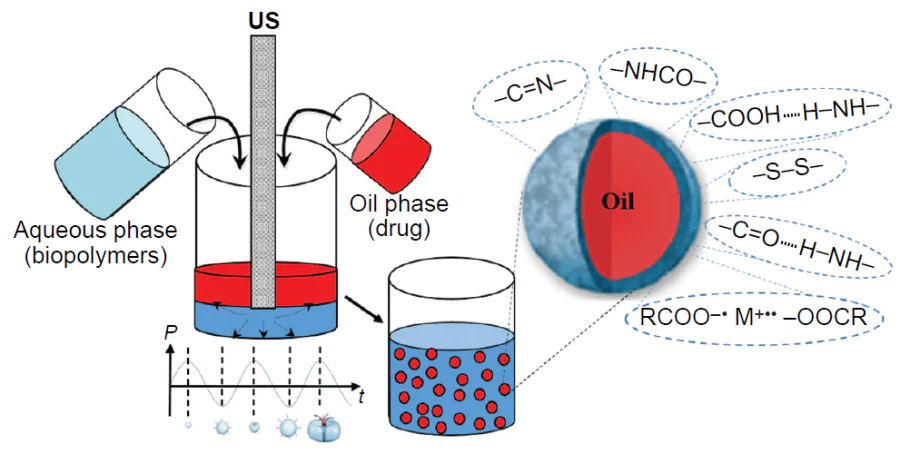
The ultrasonic emulsification is a highly effective physical method for dispersion of immiscible liquids with the aid of the acoustic cavitation phenomenon. Low-frequency ultrasound (frequency range of 20 – 100 kHz) generates variable pressure regions in a liquid medium, giving rise to cavitation bubbles (Fig. 1), the volume of which fluctuates, being increased during the expansion phase of the acoustic wave to a somewhat greater extent than it is decreased during the compression phase. Thus, the bubble volume gradually increases to reach a certain critical size. This size is called the resonant size and depends on the properties of the liquid and the ultrasound frequency. For example, at a frequency of 20 kHz in water, this size is approximately 170 μm.[17] After the resonant size has been reached, the bubble is no longer able to effectively absorb energy and becomes unstable. Under the pressure of the surrounding fluid, the cavity rapidly collapses: an implosion occurs. Exactly at this instant, conditions favourable for sonochemical reactions appear.
The collapse of cavitation bubbles generates extreme local conditions, that is, temperatures of up to 5000 K and pressures of more than 100 MPa, with the temperature variation rate being dT/dt ~ 1010 K s–1.[8][18][19] These parameters initiate a series of hydrodynamic phenomena, including the formation of shock waves and microjets, which ensure intense dispersion of the phases. It is noteworthy that the resonant size of the cavitation bubble in aqueous media (approximately 102 μm) exceeds the size of capsules being formed (0.1 – 10 μm), which indicates indirect influence of cavitation through shock waves and microjets.
The formation of capsules induced by ultrasound can be divided into two main stages.[20][21]
1. Primary dispersion: in the first stage, capillary waves develop at the interface under the influence of acoustic vibrations, leading to deformation and fragmentation of the interface with the release of macroscopic droplets of the dispersed phase into the dispersion medium. The energy efficiency of the process substantially increases when a coarse emulsion is initially formed by mechanical methods.[22][23]
2. Cavitation-induced fragmentation: in the second stage, cavitation bubbles collapse near the droplet surface and thus generate intense shock waves and microjects, which break larger droplets into submicron-sized ones.[22]
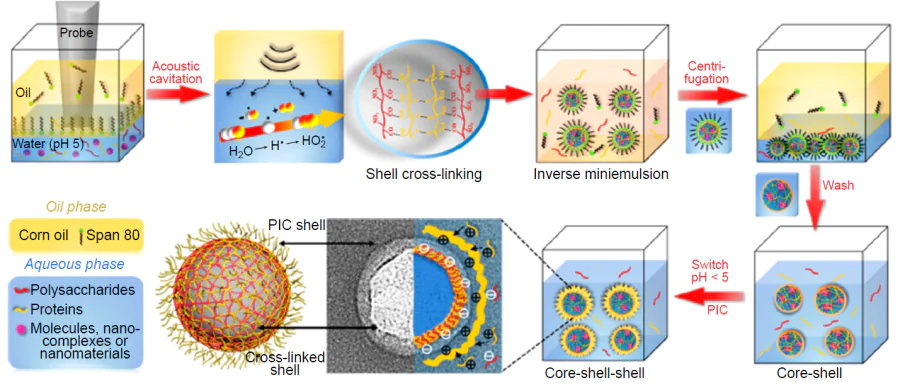
The synthesis of capsules using low-frequency ultrasound is performed in one step and does not require complicated equipment: an ultrasonic generator is mounted at the interface between oil (hydrophobic functional compound) and water (aqueous solution of a polymer) and the system is exposed to low-frequency ultrasound. When the ultrasound source is switched on, intense cavitation processes occur in the system. The energy released by collapsing cavitation bubbles not only disperses the oil, but also induces conformational changes (unfolding) and radical-induced cross-linking of macromolecules, and may also lead to the rupture and rearrangement of polymer molecules on the surface of the droplets. This gives rise to a strong, structured interfacial layer, which effectively prevents coalescence. The resulting capsules are separated and washed with water using alternating centrifugation and resuspension procedures, with this cycle being repeated until the system is completely free of excess polymer that has not been incorporated into the shell.
This procedure can be used to encapsulate a broad range of hydrophobic functional compounds, in particular air-filled polymer microspheres were prepared.[24] If further functionalization of the capsules is required, their surface is modified, for example, by electrostatic adsorption of an oppositely charged polyelectrolyte followed by centrifugation and resuspension of the system to wash away the excess polymer.
3. Mechanism of the capsule shell formation under the action of low-frequency ultrasound
3.1. The capsules with protein or polypeptide shells
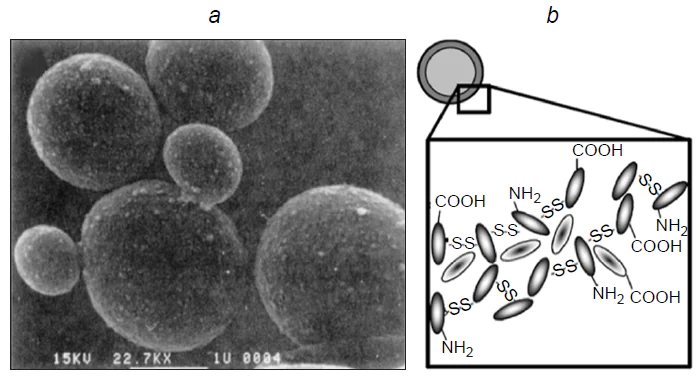
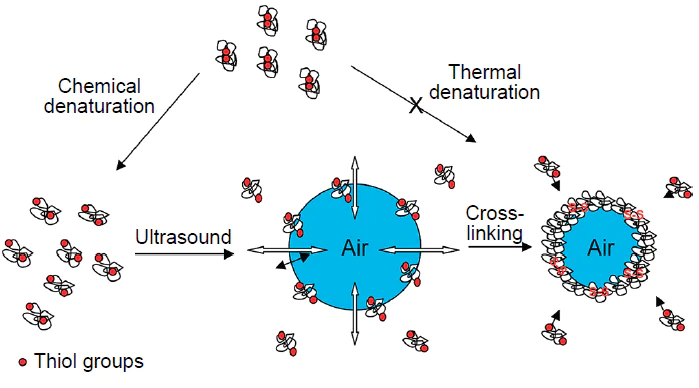
In early studies dealing with the fabrication of ultrasonic capsules, proteins were used for the formation of the polymer shells. Back in 1986, Keller et al.[25] showed that ultrasonic treatment of an aqueous solution of human albumin affords stable, air-filled albumin microspheres with a diameter of up to 10 μm. These microspheres can be used as contrast agents for imaging in ultrasound examination. The sonochemical method for the fabrication of capsules with liquid hydrophobic contents was first implemented by Suslick and Grinstaff.[5] Bovine serum albumin (BSA) microcapsules filled with n-dodecane, n-decane, n-hexane, cyclohexane, or toluene were synthesized using low-frequency ultrasound (Fig. 2a). Air-filled protein microspheres were also fabricated using BSA, human serum albumin (HSA), or haemoglobin.[24][26] It was demonstrated that mechanical homogenization alone is insufficient for the formation of stable capsules. Study of the role of oxygen in the sonochemical synthesis showed that stable microcapsules are formed when the reaction mixture is purged with air or O2, while the reaction carried out under inert atmosphere (when the solution is purged with He, Ar, or N2) does not give microcapsules.[5] Thus, thermal denaturation cannot account for the formation of stable systems.
As noted above, ultrasonic treatment of a solution gives rise to the OH• and H• radicals, the reactions of which give rise to H2 and H2O2, and, in the presence of O2, also the superoxide (O2–).[10] The hydroxyl radical, superoxide, and peroxide are potential cross-linking agents. In order to identify the role of a particular oxidant, the formation of microcapsules was studied in the presence of radical scavengers, including glutathione (a nonspecific scavenger), catalase (which decomposes hydrogen peroxide into oxygen and water), and superoxide dismutase (which decomposes superoxide into oxygen and hydrogen peroxide).[5] The results showed that the formation of capsules was inhibited by glutathione and superoxide dismutase, but not by catalase. Therefore, the authors concluded that it is superoxide that is involved in the formation of capsules as the oxidant. They assumed that superoxide accomplishes cross-linking of protein molecules through the formation of disulfide bonds between the SH groups. This hypothesis was confirmed in several studies.[24][26] The cysteine residue, which can be oxidized by superoxide, is present in BSA, HSA, and Hb, but is absent in myoglobin. In the case of ultrasonic irradiation of myoglobin solutions, the total volume of the formed microcapsules was considerably lower than that in the case of Hb.[5] It was also found that the addition of dithioerythritol (a reagent that cleaves disulfide bonds) leads to the destruction of Hb and BSA microcapsules. Finally, the oxidation of cysteine residues can be inhibited by alkylation with N-ethylmaleimide, and it has been shown that microsphere formation in solutions of alkylated haemoglobin is markedly reduced.[26] Thus, the mechanism of formation of emulsion capsules with BSA, HSA, and Hb shells under ultrasonication includes the cross-linking of macromolecules through the formation of intermolecular disulfide bonds induced by sonochemically generated superoxide (Fig. 2b).[26][27]
However, are free SH groups the only groups that form stable shells under ultrasonic irradiation? Cavalieri et al.[28] carried out ultrasonic synthesis of air-filled stable microspheres composed of chemically reduced lysozyme (Fig. 3). It was shown that stable microspheres are not formed without chemical reduction of the enzyme. The degree of molecular cross-linking was controlled by varying the degree of chemical reduction of the intramolecular disulfide bonds of the protein and the duration of ultrasonication. The microspheres remained stable for several months, with the enzymatic activity of lysozyme being retained.
Unlike lysozyme, avidin was shown to be capable of forming air-filled microspheres under ultrasonication via the formation of intermolecular disulfide bonds in the absence of free thiol groups.[12] This protein was chosen as a model for two reasons: first, avidin contains one intramolecular disulfide bond without having free SH groups; second, the well-known biological activity of avidin (biotin binding) serves as a convenient measure of preservation of the native structure. A two-stage mechanism of microsphere formation was proposed and experimentally confirmed. In the first stage, ultrasound causes cleavage of the intramolecular disulfide bridge, leading to a partial unfolding of the protein molecule. The second stage involves the formation of new intermolecular disulfide bonds between adjacent protein molecules, which provides the formation of a stable three-dimensional structure of the microsphere shell. The biological activity of avidin was preserved within the shell. Unlike conventional denaturation, which results in complete loss of protein function, the proposed sonochemical process decreases the ability of avidin to bind biotin only to a certain extent.[12] The obtained results are methodologically important, as they demonstrate the possibility of controlled transformation of protein molecules into microspheres with partial retention of the biological activity.
Some studies have shown that not only thiol groups are capable of forming stable capsule shells under ultrasonication.[13][29] Avivi and Gedanken[13] successfully used sonochemical approach to prepare emulsion microcapsules based on streptavidin, a protein devoid of free SH groups. In this case, stable microspheres were formed only in an acid medium at pH values below 6.0. The following explanation associated with a change in the protein conformation under the action of ultrasound was proposed for this phenomenon. The ultrasonication induces partial denaturation of streptavidin, which results in the exposure of hydrophobic amino acid residues that are normally buried inside the protein globule. In acid media (pH < 6.0), the carboxyl groups of the protein are protonated, which reduces their negative charge and decreases the hydrophilicity of the molecule. As a result, hydrophobic interactions start to play a major role: the protein molecules are aggregated, thus forming compact structures to minimize the contact of hydrophobic sites with water. The proposed mechanism was verified by investigation of microsphere formation from polyglutamic acid. It was found that polyglutamic acid forms capsules only at pH values below 4.5, which is close to pKa of the carboxyl side groups (~ 4.3). This confirmed the assumption proposed previously: the protonation of carboxyl groups in acidic solutions not only decreases the solubility and promotes the aggregation of protein molecules in solution, but also changes their surface properties. The formed amphiphilic aggregates are effectively adsorbed at the interface. The adsorption is energetically favourable, as this allows hydrophobic clusters to be immersed into the oil phase, thus minimizing the contact with water, which ensures the formation of a stable shell. The ultrasonication enhances both the aggregation stage and the subsequent transport of aggregates to the droplet surface.[13]
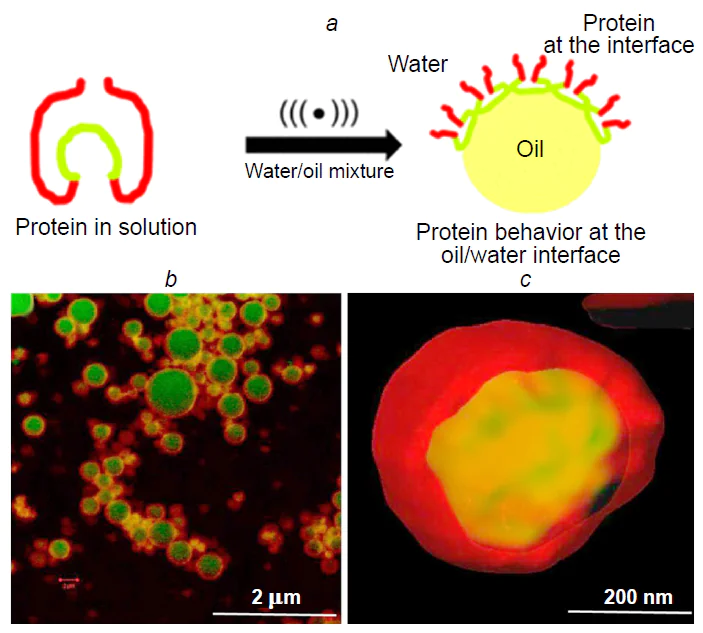
For gaining understanding on the contribution of hydrophobic interactions to the mechanism of sonochemical synthesis, it is of interest to consider experiments using a series of synthetic hexapeptides with the general formula GXGXGX, where G is glycine and X is another amino acid (alanine, serine, leucine, or aspartic acid).[29] Stable microspheres are formed only in the presence of peptides containing hydrophobic amino acid residues such as leucine. This result confirms the key role of hydrophobic interactions in the self-assembly of protein shells. The successful preparation of microspheres from proteins and polypeptides containing no cysteine residues indicates the existence of alternative particle stabilization mechanisms unrelated to the formation of disulfide bonds. In this case, the formation of microspheres can be viewed as a multi-stage process initiated by ultrasound. In aqueous solutions under native conditions, proteins usually exist as stable three-dimensional structures in which hydrophobic amino acid residues are buried inside the globule, while hydrophilic groups are located on the surface. The ultrasonication of water/organic solvent two-phase systems induces the following key transformations: first, shear forces cause partial denaturation of protein molecules, leading to the exposure of previously buried hydrophobic regions; second, modified protein molecules migrate to the interface; third, self-assembly of the shell takes place, with hydrophobic domains being directed toward the organic phase and hydrophilic ones pointing toward the aqueous phase (Fig. 4). This mechanism explains the formation of stable microspheres with an organized shell structure.
It was shown[29] that hydrophobic interactions make a relatively large contribution to the formation of a stable shell consisting of molecules that form disulfide bonds under ultrasonication. For example, the authors fabricated BSA capsules, in which Nile Red, a lipophilic fluorescent dye exhibiting solvatochromism, was used to vizualize the hydrophobic region; this made it possible to determine the local polarity of the medium from the shift of spectral bands. The confocal microscopy data (Fig. 4b,c) revealed the presence of hydrophobic regions in the protein shell, which were predominantly located in the contact areas with the oil phase. Gülseren et al.[30] proved that the surface activity of BSA molecules in an aqueous solution increases under the action of ultrasound.[30] After 45 min of ultrasonication, the surface hydrophobicity of BSA globules increased by 24%, which was due to exposure of previously buried hydrophobic amino acid residues.
Using a set of modern physicochemical methods, Doering et al.[31] proposed detailed mechanism of the formation of BSA capsule shells induced by ultrasound. It was emphasized that the native BSA molecule contains only one free SH group, while possessing 17 stable intramolecular disulfide bridges that determine its tertiary structure. The results of polyacrylamide gel electrophoresis demonstrated the formation of high-molecular-weight oligomeric BSA species in the microcapsule shells. These data were confirmed by Raman spectroscopy, which revealed characteristic changes in the secondary and tertiary protein structures. Further studies of BSA solutions sonicated under similar conditions by dynamic light scattering revealed the formation of stable protein oligomers. This confirms the ability of ultrasound to initiate the association of protein molecules, which is the key step preceding the adsorption and cross-linking of the molecules at the interface. On the basis of these results, the following molecular mechanism for the shell formation was proposed. The ultrasonication induces partial reduction of the intramolecular disulfide bonds in the BSA molecule. The new free SH groups formed in this reaction (in addition to the single native thiol group) are oxidized by superoxide radicals that are generated during ultrasonication. The oxidation generates new intermolecular disulfide bridges between neighbouring protein molecules, thus forming a branched cross-linked network. An additional stabilization of the three-dimensional structure of the microcapsules is provided by hydrogen bonds and hydrophobic interactions between protein molecules.
A special place among the studies considered in this Section belongs to the study by Suslick and co-workers,[11] who produced ultrasonic microspheres from sodium polyglutamate via electrostatic interactions and hydrogen bonds. The resulting capsules (Fig. 5) showed stability over broad ranges of pH (4.8 – 12) and temperature (20 – 64°C). To elucidate the nature of the stabilizing interactions in the system, an integrated analysis was carried out using modern physicochemical methods. Matrix-assisted laser desorption/ionization mass spectrometry did not reveal the formation of dimers or oligomers after ultrasonication, which ruled out the possibility of covalent cross-linking of polymer molecules. The addition of radical scavengers to the reaction mixture before ultrasonication did not affect the number of microspheres, thus confirming the absence of a radical stabilization mechanism. The data of IR spectroscopy showed a considerable shift of the asymmetric vibrational band for the carboxyl group from 1554 cm–1 in the solution to 1608 cm–1 in the microspheres (Δν = 54 cm–1), indicating a change in the type of interactions of carboxyl groups. These results were used to propose a mechanism for stabilization of microspheres via the formation of a hydrogen bond network between the carboxyl groups of neighbouring macromolecules, formation of [RCOO–∙∙∙M+∙∙∙–OOCR] ion pairs, where M+ is either H+ or Na+, and electrostatic interactions between ionized groups of the polymer. The proposed mechanism of the microsphere formation via non-covalent interactions accounts for their stability combined with the possibility of reversible disaggregation upon a change in the surrounding conditions.
3.2. The capsules with polysaccharide and/or glycoprotein shells
The use of proteins is not always cost-effective, and they rapidly decompose in the body via enzymatic degradation. In view of these limitations, alternative approaches to the production of emulsion capsules have been proposed, which expanded the range of compounds applicable for the formation of shells by the use of polysaccharides, glycoproteins, or mixtures of the compounds with proteins.
By analogy with protein shells, polysaccharide shells of emulsion capsules can also be formed using covalent cross-linking through the formation of disulfide bonds. For example, this approach was utilized to fabricate microcapsules consisting of chemically modified chitosan.[32] Thiol groups were introduced into chitosan structure using DL-N-acetylhomocysteine thiolactone (AHT). Microcapsules with AHT-modified chitosan shells were synthesized under ultrasonication (Fig. 6). The formation of the microcapsules included three key stages: emulsification of a non-aqueous phase to form microdroplets dispersed in an aqueous solution; adsorption of AHT–chitosan at the oil/water interface; the formation of disulfide cross-links between polymer molecules under the action of superoxide generated by ultrasound. For comparison, capsules made of non-modified chitosan were prepared. Experiments showed that the thickness of the microcapsule shell increased with increasing concentration of thiol groups from 148 ± 2 nm for the non-modified polymer to 180 ± 3 nm for AHT-modified chitosan. Atomic force microscopy study indicated that AHT-modified chitosan shells have higher stiffness (Young’s modulus of 1.44 MPa) compared to non-modified analogues (Young’s modulus of 0.83 MPa), which is indicative of enhanced mechanical strength. A similar approach based on the ultrasonic cross-linking of thiol groups was successfully applied to produce capsules composed of other thiolated polysaccharides including alginate,[33] starch,[34] carboxymethyl cellulose,[35] and hyaluronic acid.[36]
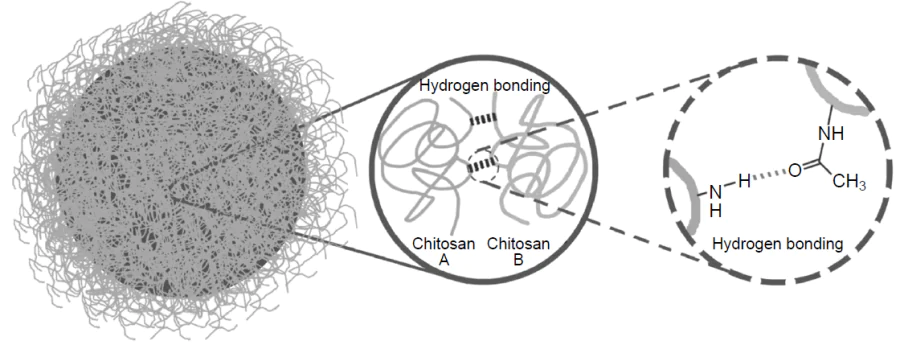
Chitosan is a polysaccharide used most commonly for the production of microcapsules, including emulsion microcapsules. The mechanism of chitosan shells formation under the ultrasound action in the case of using non-thiolated molecules is the subject of quite a few studies.[37-39] Thus, the key role of amino groups in the formation of chitosan capsules was demonstrated.[37] According to experiments, preliminary modification of chitosan with fluorescein isothiocyanate via the Schiff reaction with the polymer amino groups completely suppressed the formation of capsules during subsequent ultrasonication. The IR spectroscopy data ruled out the possibility of covalent cross-linking in the microcapsule shell under ultrasonication, since no marked changes in the spectra were observed, except for a slight (5 cm–1) shift of the intense band at 1648 cm−1 corresponding to the C=O bending mode of the acetamide groups. These groups are present in chitosan due to incomplete deacetylation of the original chitin. The authors suggested that the observed shift is attributable to the formation of hydrogen bonds between amino and acetamide groups, which are responsible for the formation of capsules (Fig. 7). The ultrasonication plays a key role in this process: first, it breaks intramolecular hydrogen bonds, thus releasing functional groups for intermolecular interactions. This mechanism is similar to the formation of avidin capsules, in which ultrasound first breaks the disulfide bonds (S – S) and then promotes their reorganization.[12] Zhu et al.[38] found more evidence for the importance of hydrogen bonds for the stability of chitosan shells.[38] The authors investigated the effect of the degree of chitosan deacetylation (content of acetamide groups) on the stability of highly concentrated emulsions (with a volume fraction of the internal phase ≥ 0.74). It was found that a decrease in the degree of deacetylation from 90% to 50% was accompanied by increasing emulsion stability, which attests to a contribution of intermolecular hydrogen bonds between the amino and acetamide groups to stabilization of the chitosan shell. These results indicate that hydrogen bonds are the key factor ensuring the formation and stability of chitosan microcapsules.
Skirtenko et al.[39] proposed an alternative mechanism for the formation of chitosan microcapsules based on the formation of covalent imine bonds (–C=N–). Using IR spectroscopy, the authors detected a characteristic shift of the absorption peak from 1645 to 1647.6 cm–1 on going from free chitosan to chitosan microspheres. The authors interpreted this shift as evidence for the formation of imine bonds between the free amino groups of chitosan and the aldehyde residues of the chitosan terminal groups. It is noteworthy that the proposed mechanism does not rule out the involvement of other types of intermolecular interactions. The authors emphasized that the formation of microspheres is apparently caused by a combination of several factors: formation of intermolecular imine bonds and hydrogen bonds (both inter- and intramolecular ones), van der Waals contacts, hydrophobic effects, and electrostatic interactions. This set of interactions accounts for the stability of the resulting structures and is in line with experimental data. The proposed mechanism complements the existing models of the formation of chitosan capsules, as it offers a more comprehensive interpretation of their structural organization and stability. The results of the study indicate that, depending on the synthesis conditions, different types of intermolecular interactions may predominate.
It is worth noting that minor shifts in the IR spectra should be interpreted with caution. As noted above, Wang et al.[37] considered the shift of the band for acetamide groups (~ 5 cm–1) to be too small to draw a conclusion about the presence of covalent cross-linking. Meanwhile, Skirnenko et al.[39] interpreted a comparable shift (~ 2.6 cm–1) in the same spectral region as being indicative of the possible formation of imine bonds. Thus, small shifts in the IR spectra cannot serve as either decisive proof or refutation of covalent cross-linking; their interpretation should be confirmed by the results of a more comprehensive study, which may include X-ray photoelectron spectroscopy and solid-state NMR spectroscopy.
In a later study, Ye et al.[40] reported additional evidence for the covalent nature of cross-linking in the shells of chitosan microcapsules, the SEM images of which are shown in Fig. 8. Experimental studies of the mechanical properties of the capsules were carried out by atomic force microscopy: the average stiffness of the microcapsules varied depending on the shell thickness from 82 mN/m (the average shell thickness of 134 ± 10 nm) to 111 mN/m (the average shell thickness of 150 ± 24 nm).[41] The application of a certain force to the capsule with monitoring of the probe position made it possible to observe a characteristic time relaxation of the force described by an exponential dependence.[40] This relaxation behaviour is typical of systems with covalent bonds.[42]
The above data indicate that the mechanism for the formation of chitosan shells is still a debatable issue. Using spectroscopic methods, different research groups obtain data that can be interpreted in favour of either mainly non-covalent (hydrogen bonds) or mainly covalent (imine bonds) cross-linking. Apparently, this points to the hybrid nature of the interactions in the shell, in which the relative contributions of different types of bonds may vary depending on the synthesis parameters.
Apart from polysaccharides, natural glycoprotein can also be successfully used for the formation of a polymer shell of emulsion microcapsules under ultrasonication, as was shown in relation to gum acacia.[6] Using IR spectroscopy, the authors revealed a pronounced effect of ultrasonication on the structure of gum acacia. First, changes in the 2124 cm–1 region were observed, indicating modification of the bonds in glucoprotein amino groups. Second, the C=O vibrational band (1680 cm–1) was found to shift by 50 cm–1 to lower wavenumbers, which is typical of the formation of amide bonds between the polymer functional groups. These data indicate that the formation of a strong and stable polymer capsule shell in this system is due to the covalent amide bonds formed under ultrasonication.
In addition to the formation of covalent bonds, ultrasonication of polysaccharides causes their controlled fragmentation, which makes a contribution to the formation of stable shells of emulsion capsules. For example, it was shown[43] that ultrasonication markedly improves the emulsifying properties of pectin via structural rearrangement of its molecules: the hydrodynamic diameter of the modified pectin decreases to 480.45 ± 26 nm and the molecular weight decreases to 286.37 ± 25 kDa compared to those of the native polymer (1288.97 ± 5 nm and 485.00 ± 8 kDa, respectively). The cavitation effects induce cleavage of glycosidic bonds and depolymerization, resulting in decreasing molecular weight and hydrodynamic diameter of pectin. This is favourable for better mobility of molecules in an aqueous medium and more effective adsorption at the interface. It is important that fragmentation is accompanied by a conformational rearrangement of the polymer chains, resulting in the exposure of hydrophobic groups that were previously buried in the native structure of the polysaccharide. These structural changes allow pectin to form a thicker and denser stabilizing layer on the surface of oil droplets. Another factor increasing the stability is the greater accessibility of functional groups that facilitate intermolecular interactions.
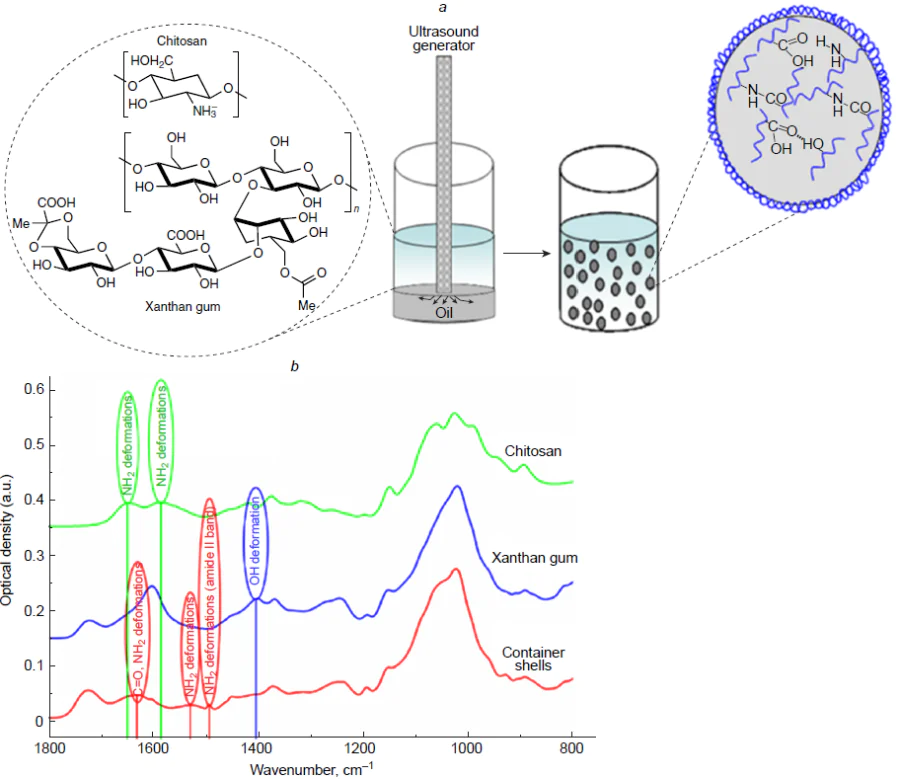
Apart from one-component systems, a mixture of two polymers able to form a polyelectrolyte complex was proposed for the ultrasound-induced formation of capsule shells. An important feature of the procedure is that ultrasonic emulsification was performed in an acid medium in which polymers did not initially form complexes. First, using this approach, highly stable capsules with chitosan and xanthan gum shells were prepared[16][44] (Fig. 9). The results of the IR spectroscopic study demonstrated the interaction between the chitosan NH2 groups and the acetate and pyruvate –COOH groups of xanthan gum, leading to the formation of amide bonds. Further development of these studies involved the replacement of chitosan by diethylaminoethyl dextran, which was approved for use in drug delivery.[45] IR spectroscopic study showed a blue shift of the absorption bands at 790 cm–1 (wagging vibration of the NH2 group), which is indicative of a contribution of the amino groups of diethylaminoethyl dextran to the interaction between polymers. In addition, the spectrum of the isolated and dried capsule shells exhibited a slight shift of the peak at 2900 cm–1 (OH stretching mode) with the appearance of a shoulder peak at 2825 cm–1, which is attributable to a contribution of xanthan gum carboxyl groups to the reaction between polyelectrolytes. Thus, changes in the IR spectra demonstrate not only the appearance of Coulomb forces between the acetate and pyruvate groups of xanthan gum and the amino groups of diethylaminoethyl dextran, but also the formation of intermolecular hydrogen and covalent amide bonds.
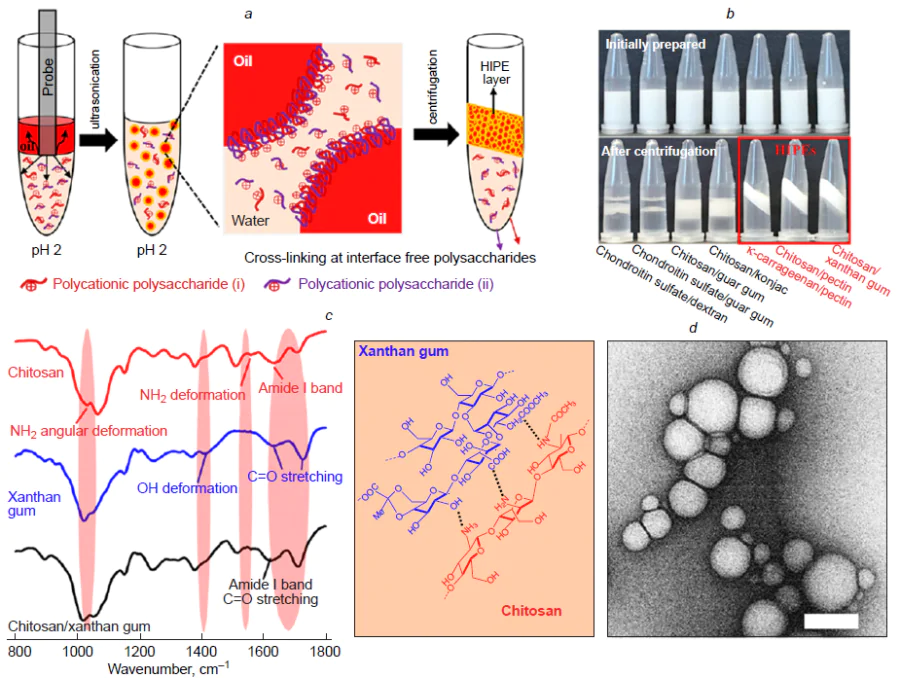
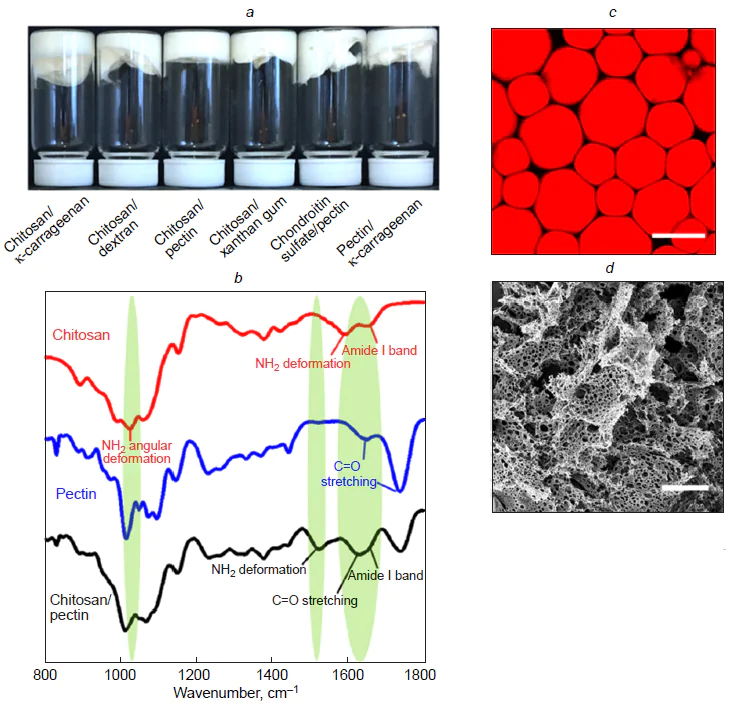
The proposed approach was extended to seven various combinations of polysaccharides used as materials for the shells for the dispersed phase (oil) droplets in order to prepare highly concentrated emulsions.[46] The following polymer pairs were chosen for the study: chondroitin sulfate/dextran, chondroitin sulfate/guar gum, chitosan/guar gum, chitosan/konjac, κ-carrageenan/pectin, chitosan/pectin, and chitosan/xanthan gum (Fig. 10a). Although all combinations formed stable emulsions after ultrasonication, only three systems, that are, κ-carrageenan/pectin, chitosan/pectin, and chitosan/xanthan gum, gave a highly concentrated layer of capsules upon centrifugation (Fig. 10b), which attests to enhanced stability of the capsules to coalescence in a centrifugal field compared to other polymer pairs. The cited authors[46] suggested that the enhanced stability may be due to the formation of stable interpolymer complexes, possibly involving covalent cross-links. However, to confirm the chemical nature of these interactions, additional spectroscopic studies were required, which actually revealed the signs of formation of amide bonds particularly in these three systems. The most characteristic changes were found for the chitosan/xanthan gum system by IR spectroscopy. The ultrasonic treatment induced a shift of the amide-I band of chitosan from 1633 to 1624 cm–1, almost complete disappearance of peaks at 1558 cm–1 (NH2 deformation vibrations) and 1031 cm–1 (NH2 angular deformation vibrations), a shift of the band for carbonyl groups of xanthan gum to lower wavenumbers (1712 cm–1), and disappearance of the peak at 1409 cm–1 (ОН vibrations of carboxyl groups) (Fig. 10c). These changes attest to the formation of amide bonds between the chitosan amino groups and the carboxyl groups of xanthan gum. Similar chemical reaction mechanisms were also established for the other two combinations, that are, κ-carrageenan/pectin and chitosan/pectin pairs, in which amino, carboxyl, hydroxyl, or sulfhydryl groups are involved in the formation of interpolymer bonds. For the other polysaccharide pairs, no significant changes in the IR spectra were observed, which accounts for the fact that they cannot form stable, highly concentrated emulsions after centrifugation. One more piece of evidence for the formation of covalent bonds between chitosan and xanthan gum in the microcapsule shell was obtained using high-performance gel permeation chromatography.[47] The chromatogram of a mixture of chitosan with xanthan gum exhibited three peaks, with the major one corresponding to polysaccharides with a molecular weight of 77.31 × 104 g mol–1. In the sonicated mixture, this value was 319.50 × 104 g mol–1. The increase in the polymer molecular weight after ultrasonic treatment is indicative of the formation of intermolecular cross-linking products.
Another study of the chitosan/pectin system[48] also demonstrated the key role of chemical reactions between components for stabilization of an emulsion. A control experiment with a mechanically homogenized mixture showed that it was unstable and rapidly separated into phases during centrifugation, whereas ultrasonication yielded stable, highly concentrated emulsions (Fig. 11a). It is important to note that neither chitosan nor pectin alone were able to stabilize such system, which confirms the necessity of interaction between the two polymers. IR spectra showed that after ultrasonication, the peak at 1558 cm−1 attributable to NH2 deformation vibrations in chitosan shifted to 1523 cm–1, while the peak at 1031 cm–1 associated with the angular deformations of the NH2 group disappeared (Fig. 11b). Meanwhile, the band corresponding to the C=O stretching vibrations of free carboxyl groups in pectin shifted to a lower wavenumber (1627 cm–1) and merged with the amide-I band of chitosan to form a broad band. X-ray photoelectron spectroscopy detected the appearance of a signature typical of the amide bond (–NHCO–). These data confirm the formation of amide bonds between the chitosan amino groups and pectin carboxyl groups in the shells of emulsion capsules prepared under ultrasonication. This covalent cross-linking markedly increases the mechanical strength and integrity of the shells of single droplets. This is manifested in the fact that the droplets in the highly concentrated dense emulsion layer formed upon centrifugation do not coalesce (Fig. 11c,d ). Thus, the cross-linked shell prevents droplets from coalescence under the influence of centrifugal forces, which is a key requirement for the formation of highly concentrated emulsions resistant to coalescence.
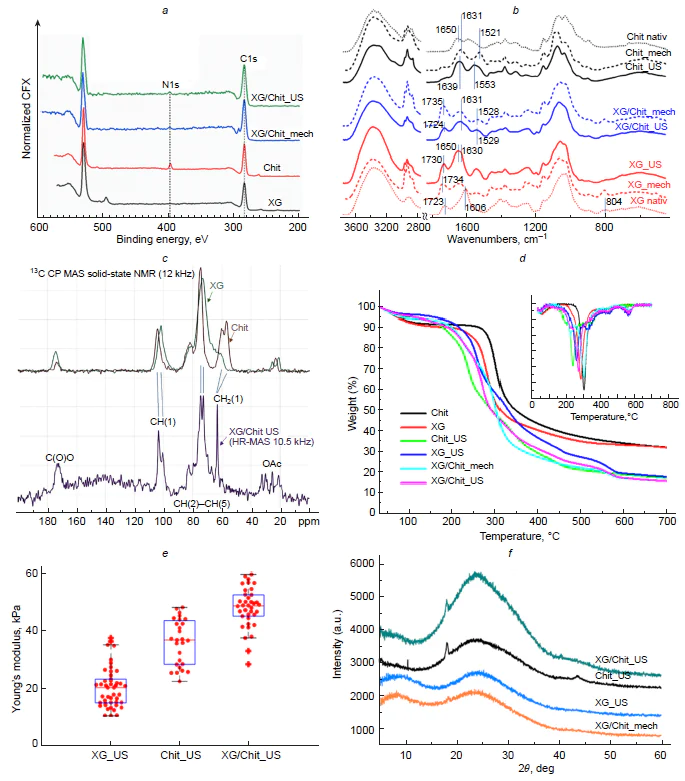
A systematic comparative study of the capsules formed upon ultrasonic treatment and mechanical emulsification using both single (xanthan gum, chitosan) and mixed polysaccharide systems demonstrated the unique influence of the ultrasonication on the structure and stability of the shells.[49] An integrated use of modern analytical methods provided convincing evidence for the formation of covalent bonds under ultrasonication (Fig. 12). X-Ray photoelectron spectroscopy data showed a characteristic shift of the N1s peak to 400 – 402 eV and a change in the N/C ratio from 1.0 (mechanical treatment) to 1.17 (ultrasonication), indicating that the shell is enriched with the xanthan gum (Fig. 12a). According to IR spectroscopy data, the formation of shells composed of chitosan, xanthan gum, and their mixture is accompanied by structural changes in the macromolecules: the shifts of the carbonyl, amide, and hydroxyl spectral bands attest to strengthening of hydrogen bonds and rearrangement of carboxyl groups (Fig. 12b). In the case of mixed polysaccharide shell, these changes point to the formation of a polyelectrolyte complex between the protonated amino groups of chitosan and the carboxyl groups of xanthan gum. Further evidence was obtained by solid-state NMR, which revealed signal narrowing, indicating the formation of an intermolecular bond between the pristine polymers within the capsule shell (Fig. 12c). The most pronounced changes in the 13C chemical shifts were observed for the peripheral primary CH2OH and CH2OAc groups, which play the most active role in the formation of the capsule shells via the formation of intermolecular bonds. The absence of initial signals for chitosan or xanthan gum attests to the formation of a polymer shell with a fairly uniform distribution of polymers, without clusters of single-component phases. The thermal properties of the capsules were studied by TGA (Fig. 12d ). The DSC curves of all samples showed three main stages: evaporation of water (30 – 100°C), destruction of polysaccharides (200 – 500°C), and carbonization (> 500°C). The ultrasonic treatment considerably modified the decomposition profile: the decrease in the destruction temperature for single-component Chit_US and XG_US capsules is indicative of depolymerization, while for the two-component capsules (XG/Chit_US), the thermogram became more complicated. The extension of the temperature range and the appearance of new peaks point to stronger intermolecular interactions and possible cross-linking of the components under ultrasonication, as opposed to the mechanical mixture XG/Chit_mech, which preserved features of the single polymers. An important reason in favour of the efficiency of the ultrasonic method was provided by a study of mechanical properties: The Young’s modulus for the mixed capsules was 48 kPa, which exceeds these values for single-component systems (20 kPa for xanthan gum capsules and 36 kPa for chitosan capsules) (Fig. 12e). These data prove that stabilization of the capsules is not merely due to the self-assembly of macromolecules at the interface, but is a direct consequence of specific effects of ultrasonication, including depolymerization, which causes molecular reorientation and gives rise to new bonds. A powder X-ray diffraction study showed a change in the crystal structure: disappearance of the reflections from the original polymers and appearance of new diffraction peaks, indicating a new type of ordering in the shell (Fig. 12f ).
3.3. The containers with protein and polysaccharide shells
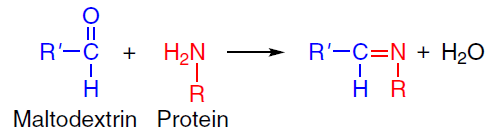
An increase in the capsule yield and stability can be achieved by using combined protein and polysaccharide systems due to the formation of additional intermolecular bonds between the components under ultrasonication. For example, in the case of the emulsion microcapsules with a shell composed of a soy protein isolate – maltodextrin complex, ultrasonication was found to improve the properties compared to those of control samples, including an increase in the uniformity of capsule size distribution (transition from bimodal to unimodal distribution), a decrease in the average droplet size from 20.5 to 10.7 μm, and increase in the efficiency of hemp seed oil encapsulation from 77% to 95%.[50] The key effect of the ultrasound was the formation of covalent bonds between the shell components by the Maillard reaction, which involves free amino groups of soy protein and the terminal carbonyl groups of maltodextrin (Scheme 1). This process is intensified owing to physicochemical conditions generated by cavitation, which promote the structural rearrangement of the protein molecules. Denaturation and unfolding of the protein globule result in exposure of previously buried amino groups, which increases the number of accessible reaction sites to react with maltodextrin. Spectroscopic studies confirmed the enhancement of covalent bonds between the components: the intensity of characteristic peaks at 1021 and 1161 cm–1 corresponding to the stretching bands of the C – O – C glycosidic bonds increased. The degree of glycosylation of the soy protein/maltodextrin complex was found to increase upon the ultrasonic treatment to reach 15.82% compared to the control sample (4.77%).
Original capsules with a water core and BSA/chondroitin sulfate (CS) shell were synthesized by inverse miniemulsion polymerization with anthocyanin as a model hydrophilic bioactive compound[51] (Fig. 13). Comparative analysis showed that the shell made of the BSA/CS combination increases the yield of the capsules by 59.9% compared to the systems contained only BSA. X-Ray photoelectron spectroscopy confirmed the formation of disulfide bonds upon the oxidation of thiol groups and the formation of amide bonds between the BSA amino groups and the CS carboxyl groups. According to XPS data, the degree of cross-linking for BSA and BSA/CS systems was 8.4 and 14.2%, respectively. Data on polyacrylamide gel electrophoresis identified the formation of high-molecular-weight moieties, indicating intermolecular cross-linking induced by ultrasound. The versatility of this approach was confirmed by successful use of other polysaccharides (κ-carrageenan, dextran sulfate, pectin, and xanthan gum) in combination with BSA. Pomon et al.[52] expanded the scope and applicability of this method by using hydrolyzed whey protein isolate and pectin to form the shell with rebaudioside A as the hydrophilic content. High-resolution X-ray photoelectron spectroscopy confirmed the formation of amide bonds between the amino groups of the protein and the carboxyl groups of both the protein and pectin.
4. Application prospects of the capsules produced using ultrasound
The ultrasonic method for the synthesis of the polymer capsules proved to be effective for encapsulation of a wide range of bioactive compounds while preserving their functional properties. Numerous studies confirmed the successful incorporation of plant extracts such as astragalus oil[53] and pandan extract[54] into the protein and polysaccharide shells, with the biological activity of the extracts being retained after encapsulation. The results of drug loading are of particular interest: micro-encapsulated antibiotic tetracycline[55][56] showed an antibacterial activity comparable to that of the free drug, while the anticancer drugs Taxol[57] and Gemzar[58] exhibited much higher anticancer activity when encapsulated in microcontainers than the free drugs in equimolar concentrations. The preservation of the biological activity of encapsulated hydrophobic functional compounds indicates that ultrasound has no detrimental effect on their properties. Using UV spectroscopy, Avivi et al.[55] showed that ultrasonication does not destroy encapsulated tetracycline.
The encapsulation process using ultrasound proved to be versatile for various classes of compounds. Successful encapsulation was reported for aromatic compounds (e.g., amyl acetate),[59] which served simultaneously as an active component and as an organic phase, for photochromic spirobenzopyrans,[60] and for biologically important RNA molecules, which were delivered using specially designed delivery agents based on BSA[61] and modified chitosan.[62] The practical use of these systems includes the development of antimicrobial agents (thymol and carvacrol encapsulated into the BSA microspheres),[63] effective sorbents for the removal of industrial dyes (rhodamines, methylene blue),[64] and tissue-specific drug delivery systems.[65]
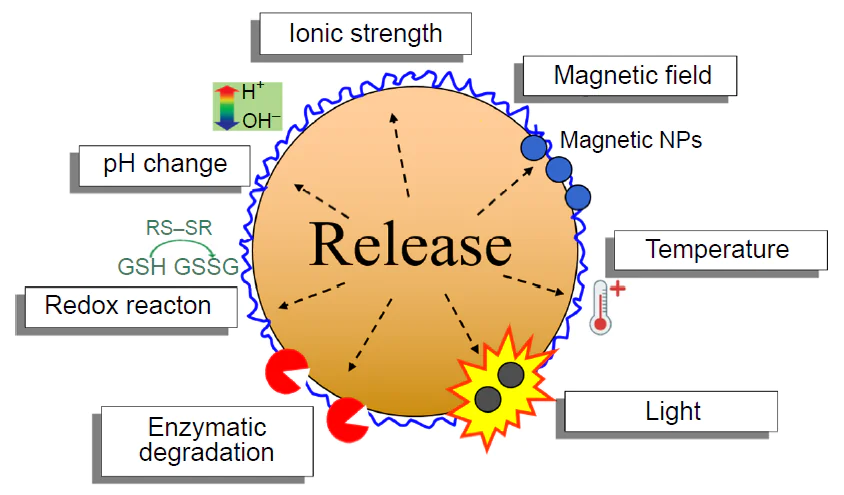
The sonochemical method offers broad potential for the encapsulation of not only hydrophobic, but also hydrophilic compounds using inverse emulsions.[66][67] Air-filled microspheres with the albumin shell produced using ultrasonication are of particular interest. The commercial product Albunex[68] developed resorting to the technique first reported by Keller et al.[25] is used as an effective ultrasound contrast agent.[69] Further development of this technique resulted in the fabrication of multimodal imaging systems, that is, the albumin microspheres modified with fluorescent dyes, which enable combined ultrasound and fluorescence diagnosis.[70] A promising trend is to use these microspheres for therapeutic purposes, particularly in photodynamic therapy.[71] Bhargawa et al.[72] described an approach to the cancer treatment, where air-filled lysozyme microspheres with the shell modified by gold nanorods and 5-fluorouracil exhibited dual action: in in vitro experiments, under the action of ultrasound, controlled drug release and pore formation in cancer cell membranes took place, which markedly increased the efficiency of chemotherapy. The capsules synthesized by the sonochemical method can be used in the development of controlled drug release systems, which can be activated by both external and internal stimuli (Fig. 14).
For example, it was shown[73] that the BSA-based capsules exhibit pronounced redox sensitivity, allowing control of the rate and degree of release of the encapsulated compound by varying the concentration of the reducing agent, DL-dithiothreitol, in the dialysate (10 μM, 1 mM, 5 mM, and 10 mM). A similar mechanism was found to operate in the presence of glutathione, which acts as a natural electron donor by reducing disulfide bonds to free thiol groups.[74] These systems are particularly valuable in the context of targeted delivery of anticancer drugs, since, depending on the cell type, the intracellular medium is characterized by high concentrations of glutathione (0.5 – 10 mM),[75] whereas glutathione concentration in the blood plasma is 2.09 ± 1.14 μM.[76] It was shown that glutathione effectively induces the drug release from the microcapsules based on thiolated chitosan[77] and thiolated carboxymethylcellulose.[35] A study of the effect of the reducing agent concentration showed the release of only 25 mass % of encapsulated coumarin 6 dye within 48 hs under glutathione concentration is of 10 μM, which is comparable with the concentration in blood plasma. This attests to the system stability in the bloodstream.[35] Meanwhile, for the concentration typical of the intracellular medium (10 mM), virtually complete drug release is achieved (91.2 mass %) within 48 h. An alternative approach to controlled drug release was developed by Han et al.,[78][79] who developed thermosensitive protein containers with a gel-like core. In this system, the gelator dissolved in an oil core is self-assembled on cooling from 45°C to room temperature (25°C) to give a three-dimensional network, which induces phase transition of the whole core from a liquid state to a gel (sol – gel transition). It was shown by in vitro experiment that the subsequent heating above the sol – gel transition temperature results in the formation of a liquid core and destruction of shells during stirring, which induces complete release of the model drug (indomethacin), as opposed to the capsules with a gel-like core, which release only 15 mass % of the drug. These studies demonstrate the variety of controlled-release strategies implemented in the sonochemically produced microcapsules.
Magnetic capsules are of particular interest for targeted drug delivery, as their movement can be controlled using an external magnetic field. Various research groups developed approaches to the design of such systems. Avivi et al.[80] proposed an original integrated method combining sonochemical synthesis of nanostructured iron oxide and the formation of the protein microspheres. They used two procedures for iron introduction: the addition of iron pentacarbonyl to the organic phase and dissolution of iron acetate in the aqueous phase. An alternative strategy was developed by Han et al.,[81] who fabricated the magnetic containers by layer-by-layer deposition of Fe3O4 nanoparticles on the surface of the protein microspheres. In addition, successful sonochemical synthesis was reported for the preparation of the magnetic microspheres based on various biopolymers: BSA,[82][83] chitosan,[84] and thiolated alginate[33] with encapsulated hydrophobic Fe3O4 nanoparticles. The use of biocompatible materials (proteins and polysaccharides) in combination with magnetic nanoparticles opens up new prospects for the fabrication of effective targeted drug delivery systems.
In recent years, a number of strategies for modification of containers to improve the efficiency of targeted drug delivery have been developed. A promising line of research is the use of folic acid as a ligand, since folate receptors are overexpressed on the surface of many types of cancer cells. The ultrasonic capsules made of various materials, such as HSA,[85] starch,[34] ε-poly-L-lysine,[86] and carboxymethyl cellulose,[35] were successfully modified with folic acid. Another approach involves the use of monoclonal antibodies for the targeted delivery of the HSA capsules.[87] Skoll et al.[88] showed that incorporation of wheat germ agglutinin into the protein shell of capsules increased their binding to urothelial cells by 55% compared to non-modified samples. Toublan et al.[89] developed an electrostatic method for layer-by-layer modification of the BSA microspheres with RGD peptides. This enabled successful targeting of integrin receptors, which are overexpressed in colon tumours.[89] Three types of peptide sequences (RGDKKKKKKKKKKKKK, KKKKRGDKKKK, and KKKKKKRGD) exhibited enhanced binding to the cancer cells. Grinberg et al.[90] showed a clear-cut selectivity of the BSA microspheres containing gemcitabine and anti-EGFR antibodies to pancreatic cancer cells.
A promising area of research is the development of multimodal systems that combine magnetic properties with molecular targeting. A number of publications[77][84][91-93] described containers loaded simultaneously with magnetic nanoparticles to control their migration and location by an external magnetic field and targeted ligands (folic acid, peptides, or antibodies) for selective binding to the target cells. These combined approaches make it possible to implement the concept of magnetic targeting followed by molecular adhesion, thus markedly increasing the accuracy of therapeutic agent delivery.
5. Conclusion
The ultrasonic synthesis of polymer capsules is a rapidly developing field at the intersection of chemistry and materials science, with a key achievement of recent decades being a deep understanding of the mechanism of the process: acoustic cavitation not only provides dispersion of the phases, but also establishes extreme conditions that initiate the generation of active radicals and a set of physicochemical transformations in biopolymers. These transformations include partial denaturation of proteins to expose hydrophobic regions and depolymerization of macromolecules. Altogether, this triggers self-assembly processes and promotes cross-linking of molecules at the interface. The method proved to be versatile for encapsulation of a wide range of compounds, while the practical value of the method was confirmed by successful commercialization of ultrasonic contrast agents.
However, as in all other procedures based on emulsification and using biological compounds, the ultrasonic production of the capsules is faced with issues related to sample polydispersity and decrease in the biological activity of the components. In addition, widespread implementation of the described technique for the production of delivery systems for hydrophobic compounds, in particular drugs, requires further development of laboratory and industrial processes to scale up the synthesis techniques.
The sonochemical method, which combines a single-step process with the possibility of using biocompatible polymers, is an effective and versatile tool for the fabrication of functional nano- and microcapsules with specified properties for applications in medicine, food industry, and biotechnology. The future of this research area would involve interdisciplinary approaches and development of hybrid theranostic platforms combining delivery, imaging, and controlled-release capabilities, which will open up new horizons in personalized medicine. In addition to biomedicine, the method holds promise for the food industry, agricultural technology, cosmetology, and green chemistry as a basis for developing smart delivery systems. Thus, combining the synthesis of polymer capsules using low-frequency ultrasound with advanced industrial technologies would lay the groundwork for the production of a new generation of functional microcontainers.
This work was performed within the State Assignment of the National Research Center Kurchatov Institute.
6. List of abbreviations and symbols
AHT — DL-N-acetylhomocysteine thiolactone,
BSA — bovine serum albumin,
SEM — scanning electron microscopy,
CS — chondroitin sulfate,
HSA — human serum albumin.
References