Keywords
Abstract
The proportion of waste electrical and electronic equipment is increasing year by year. This phenomenon is driven by the economic development and growing consumption. Acrylonitrile–butadiene–styrene copolymers represent a substantial constituent of electronic and household appliance waste. Butadienenitrile rubber and polyacrylonitrile are also classified as high-tonnage products. The waste from these polymers share the presence of a nitrile moiety in the macromolecules and the impossibility of selective solvolysis/hydrolysis to obtain monomers, as in the case of polyethylene terephthalate, polyamides, and polyimides. The chemical recycling of acrylonitrile copolymers is frequently rendered more complex by the presence of brominated flame retardants and synergists and therefore the elevated levels of nitrogen and bromine compounds present in the products. The review methodically analyzes the chemical recycling of polyacrylonitrile and its copolymers, including hydrolysis, hydrothermal recycling, gasification, and pyrolysis. The primary focus of this study is on brominated waste, which poses significant challenges in terms of reuse without undergoing chemical recycling. In contrast to previous review publications, the present study provides comprehensive analysis of the hydrocarbon composition of polymer conversion products and the content of nitrogen- and bromine-containing organic compounds. The potential for the further refining of pyrolysis products to yield not only fuels but also high value-added products, such as monomers, is considered separately. The potential of methods for preliminary preparation of polymer waste for recycling including hydrolysis and hydrothermal treatment, which have not received sufficient attention previously, is demonstrated.
The bibliography includes 182 references.
1. Introduction
The modern approach to chemical recycling of plastic waste by hydrolysis involves the desire to selectively obtain products with the highest value added, such as monomers, instead of a mixture of hydrocarbons or fuels.[1-3] This approach, however, does not apply to all polymers. While polyethylene terephthalate (PET), polymethyl methacrylate (PMMA), polyamides (PA), polyurethanes, and poly-α-methylstyrene can be selectively recycled into monomers due to the presence of carbon – heteroatom bonds or quaternary carbon atoms in their backbones, and their depolymerization predominantly yields the starting monomers, acrylonitrile copolymers cannot be selectively depolymerized. In this regard, it is important to single out polyacrylonitrile (PAN) and acrylonitrile copolymers in the context of plastic waste recycling as a separate group, the chemical processing of which is inevitably associated with the production of a complex mixture of products by breaking the C – C bonds of the polymer chain.
Global production of acrylonitrile – butadiene – styrene (ABS) copolymers is growing steadily by 4 – 5% annually. In 2025, global ABS production volume was 9.75 million tons,* and by 2030, production volume is expected to increase to 12.2 million tons. The greatest growth in ABS plastic production and consumption is observed in Asian countries, particularly China. In Russia, the production volume of ABS plastic in 2023 amounted to 22 thousand tons.‡ The consumption volume was about 50 thousand tons. The global production volume of PAN in 2023 was 2.1 million tons,§ and that of nitrile butadiene rubber (NBR) in 2025 reached 1.19 million tons.¶ In Russia, 49.9 thousand tons of NBR were produced in 2022.[4] Unfortunately, it is difficult to determine the exact volume of acrylonitrile copolymer waste for several reasons: it is not always possible to separate ABS, styrene–acrylonitrile (SAN) and PAN waste from other polymer waste; not all regions of the world have established qualified collection and disposal of plastic waste, including household appliances and electronics, which are mixtures of polymers, metals, and composite materials.[5][6] Currently, about 1.6 million tons of waste electrical and electronic equipment (WEEE) is generated in Russia each year, and only about 6% is recycled.††
PAN is used to produce synthetic fibers used to make technical fabrics and knitted goods.[6][7] NBR is used in the production of cable insulation, gaskets, seals, hose-pipes, pipes, hoses, conveyor belts, fuel tanks, and as part of various composite materials.[8] SAN and ABS have the widest range of applications among polymers with a nitrile group: the automotive industry, the production of electrical and electronic products, and the construction industry.‡‡
ABS copolymer is one of the main components of WEEE.[9] Thus, the share of ABS in waste monitors reaches 69%, in printing and copying equipment it is 31%, in mobile phones, laptops, tablets it is 28%.[9][10] Other polymers that make up the WEEE composition include polystyrene (PS), polycarbonate, polyvinyl chloride (PVC), PMMA, SAN, polypropylene (PP), polyethylene (PE).
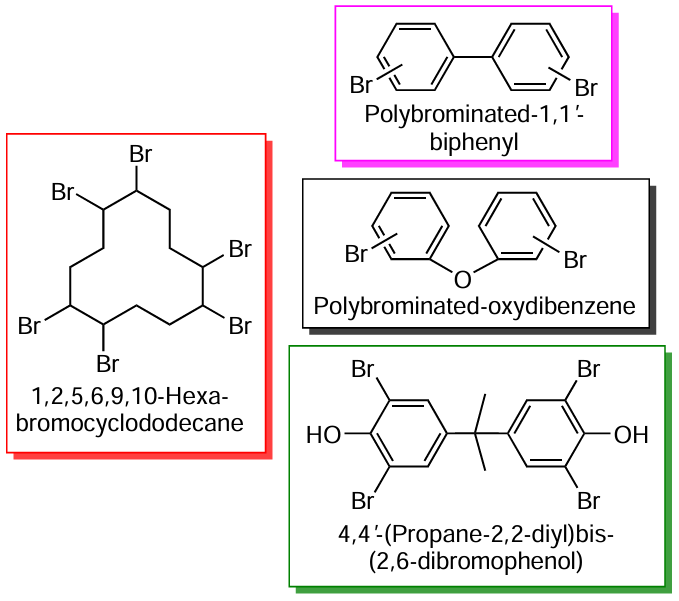
In addition to the above-mentioned polymers, WEEE contains plasticizers, stabilizers, dyes, and flame retardants, usually those bromine-containing,[9] which include four classes of compounds: polybrominated biphenyls, polybrominated diphenyl ethers, tetrabromobisphenol A, hexabromocyclododecane.[11][12] Mechanical recycling of ABS, due to the presence of brominated flame retardants and additives of metal compounds,[13] is often impossible or limited to four cycles.§§ The number of mechanical recycling cycles can be increased by the non-use of brominated flame retardants, compounds, and composites, as well as by observing labeling indicating the category and characteristics of the polymer and changing the approach to product design to facilitate the separation of materials. The already accumulated volume of WEEE, including ABS and SAN, requires chemical recycling, the main difficulty of which lies in the presence of N and Br atoms in the plastic mixture supplied for recycling and, accordingly, in the recycling products.
Methods for recycling plastic mixtures that make up WEEE, including mechanical recycling and recycling strategies (i.e., returning to the life cycle without chemical recycling), debromination, chemical recycling, viz., pyrolysis, hydrothermal liquefaction, gasification, are covered in a number of reviews.[10][12-16] The possibility of using plastic waste in 3D printing is analyzed.[17] Reviews[14][16] pay considerable attention to the possibilities of recycling as the most economically and environmentally attractive trend for ABS recycling; in particular, flotation separation methods for polymers and extraction methods for separating polymer mixtures are considered. Reviews[5][6][13] address the challenges of WEEE recycling. However, these reviews do not highlight the peculiarities of hydrolysis, hydrothermal transformations, and pyrolysis of pure ABS, SAN, PAN, and NBR. Thus, to date, no review has compiled and analyzed all the nuances of recycling nitrile-containing polymers.
In this review, chemical recycling routes for nitrile-containing polymers into feedstocks for petrochemicals and fuels are analyzed. Special attention is paid to the composition of the recycled products, including the content of organonitrogen, -chlorine, and -bromine compounds, with the aim of further developing product refinement technologies to produce not only fuels but also high value-added products, which have not been analyzed in previous reviews. We also tried to pay special attention to the methods of preparation for chemical recycling of acrylonitrile copolymers (including those containing brominated flame retardants), including denitrogenation and debromination, which was also not covered in other review publications. Since ABS plastic, which does not contain flame retardant additives, can be easily recycled and does not require chemical processing,[14][16][17] this review focuses on brominated ABS and SAN waste, the reuse of which without chemical recycling is difficult.
* https://www.mordorintelligence.com/industry-reports/acrylonitrile-butadiene-styrene-abs-resin-market [accessed date 02.02.2026]
‡ https://oaoplastic.ru / [accessed date 02.02.2026]
§ https://www.marketreportsworld.com/market-reports/polyacrylonitrile-market-14717429 [accessed date 02.02.2026]
¶ https://www.mordorintelligence.com/industry-reports/nitrile-butadiene-rubber-market [accessed date 02.02.2026]
†† https://reo.ru/tpost/oac9fp5b61-chislo-punktov-po-priemu-othodov-elektro [accessed date 02.02.2026]
‡‡ https://www.gminsights.com/industry-analysis/acrylonitrile-butadiene-styrene-ABS-market [accessed date 02.02.2026]
§§ https://reo.ru/tpost/mloypfeso1-pererabotka-othodov-abs-plastika-v-rossi [accessed date 02.02.2026]
2. Features of acrylonitrile copolymers hindering their return to the life cycle without chemical recycling
First of all, the main trends in modern research in the field of polymer waste recycling should be identified. These are recycling after separation and purification from impurities;[11][13][18-23] chemical functionalization or modification of polymer waste without breaking the polymer backbone in order to produce materials with better or unique physical and mechanical properties;[24] the use in 3D printing;[14][25][26] chemical recycling with selective production of monomers or other valuable chemical products.[1][3][10][12-16][27] Accordingly, non-selective chemical recycling to obtain fuel components is undesirable due to the need for multistep refining of the latter and low demand for such fuels.
The bromine content in ABS plastic used for the production of household appliances and electronics can amount to 10 wt.%.[9][11][12] Together with brominated flame retardants, Sb2O3 as a synergist is always used, the role of which is to form SbBr3 , SbOBr and Sb4O5Br2 in the course of the thermal destruction of the flame retardant during the combustion of the polymer. These species react with free radicals responsible for the spread of flame, breaking the chain reaction.[28]Scheme 1 shows the main types of available brominated flame retardants.
It has been proven[11] that brominated flame retardants in polymer waste can undergo thermal degradation under the action of solar radiation, releasing volatile bromine-containing compounds. This is one of the reasons making the recycling of brominated polymers challenging. Another reason is the heterogeneity of the composition of different types of waste (various contents of bromine atoms, synergists, different types of additives, etc.), which significantly degrade the characteristics of the resulting polymer products.[22][23] Therefore, chemical recycling of such polymers is more feasible.
Technologies for chemical recycling of polymer waste can be classified as non-selective (pyrolysis, hydroconversion, gasification, hydrothermal liquefaction) and selective (hydrolysis, solvolysis).[10][12-16][27-32] The latter involve selective depolymerization and are applicable to polymers containing C – X (X = N, O) bonds in the main chain: polyesters, polyurethanes, polycarbonates, and polyamides. A comprehensive review[32] has been devoted to the selective chemical recycling of polymer waste. It is also obvious that selective methods are not applicable to the recycling of acrylonitrile copolymers due to the absence of C – X bonds in the backbone, so we will not dwell on them here.
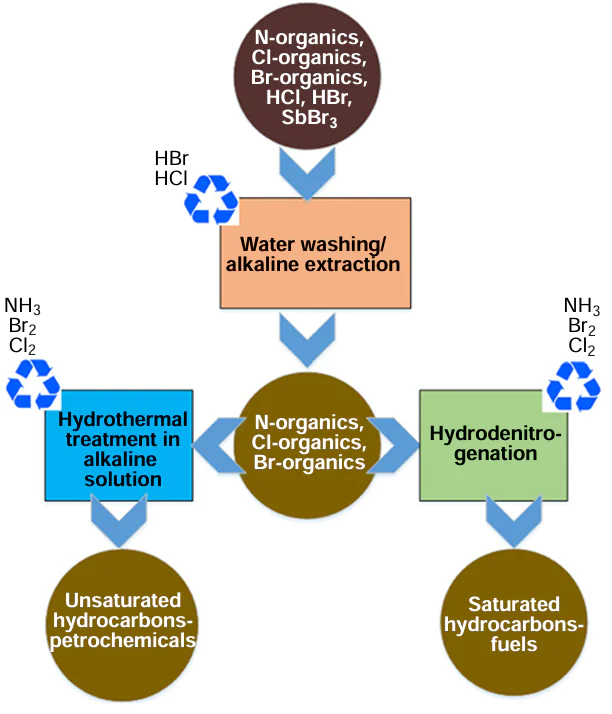
While the pyrolysis of polyolefins produces a mixture of hydrocarbons, the pyrolysis of, e.g., ABS plastic produces a mixture of hydrocarbons, nitrogen, and organobromine compounds. Therefore, acrylonitrile copolymers containing brominated flame retardants can be considered among the most ‘challenging’ polymer wastes from the perspective of a ‘green’ and circular economy approach.[33-35] In this regard, the preparation of plastic waste for recycling, combining denitrogenation and debromination,[36] as well as the purification and separation of the products of the main processing stage (pyrolysis, hydroconversion, or hydrothermal liquefaction), is of particular importance. This review is structured in such a way that it first discusses methods for preparing plastic waste for recycling, then the main types of non-selective chemical recycling, and finally, product refining technologies.
3. Preparation for recycling by hydrolysis
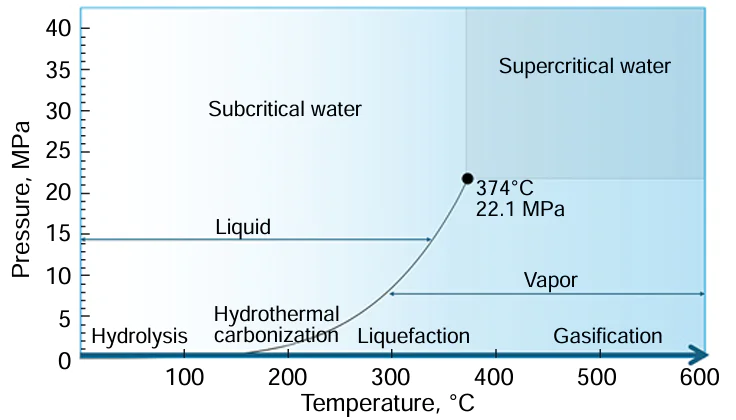
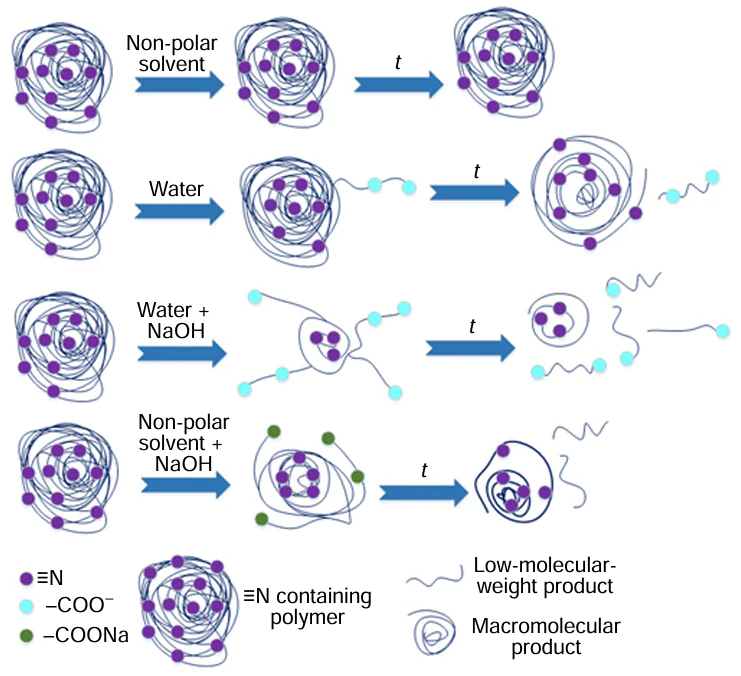
First of all, it is necessary to clarify the differences between hydrothermal recycling and hydrolysis of plastic waste. Fig. 1 shows a phase diagram of water indicating the approximate temperature ranges for hydrolysis, hydrothermal carbonization, liquefaction, and gasification processes.[1] Hydrolysis is carried out at moderate temperatures (from room temperature to approximately 200°C). The main difference between the hydrolysis of polymer macromolecules and hydrothermal recycling is that during hydrolysis, only functional groups (nitrile, amide) undergo transformations, while the polymer hydrocarbon backbone remains practically intact, whereas hydrothermal recycling involves free-radical processes of destruction of the polymer backbone.
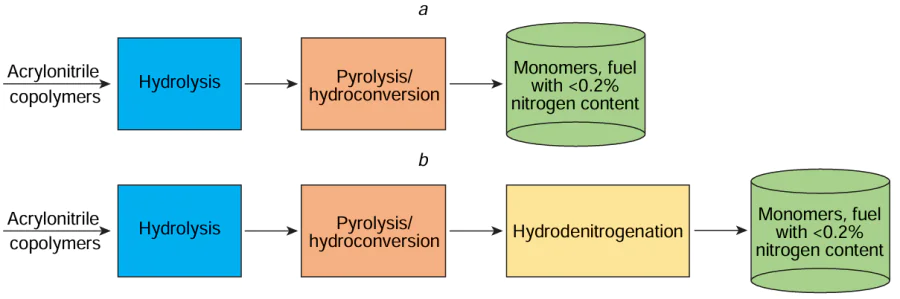
With regard to the recycling of nitrile-containing plastic waste, hydrolysis can be considered as the first step of a two-step process[37] (or a three-step process with the addition of a hydrodenitrogenation step, Fig. 2), aimed at reducing the N content in polymer macromolecules in order to minimize the formation of undesirable nitrogen-containing organic products in the second step of recycling (pyrolysis, hydrothermal recycling, hydrocracking). Therefore, carrying out hydrolysis, a low-cost and technologically simple process, ensures the production of a product with a low N content through the subsequent pyrolysis, which significantly simplifies its further refining or makes it possible to use it as a fuel, and also reduces corrosion of equipment by nitrogen compounds in the pyrolysis step.
Hydrolysis of PAN is a well-studied and long-known process used to obtain and utilize partially hydrolyzed polymers, the macromolecules of which have some of the nitrile groups replaced by carboxyl moieties, as flocculants or components of drilling fluids and coatings.[38][39] Recently, interest in hydrolyzed PAN used as a component of membranes has increased.[40][41] A distinction is made between hydrolysis of PAN in acidic, neutral, and alkaline medium. Acid hydrolysis is typically carried out in HNO3 or H2SO4 solutions.[38][42][43] In this case, as shown on the example of PAN hydrolysis in 65% HNO3, nitrile groups are almost completely converted into CONH2 moieties and only partially into COOH moieties.[42][43] Acid hydrolysis cannot be considered as an option for preparing plastic waste for further recycling, primarily due to the ineffective removal of nitrogen atoms, as well as because of the use of concentrated acid solutions, which is economically and technologically impractical and environmentally hazardous.
Hydrolysis of PAN in a neutral media has been understudied and is rarely performed, as achieving a nitrile group conversion above 70% requires a higher temperature than alkaline hydrolysis. Thus, hydrolysis of PAN under hydrothermal conditions at 180°C for 12 h affords hydrolyzed PAN that is free of nitrile groups, according to 13C NMR data.[44] The high degree of conversion of nitrile groups to carboxyl groups is explained by the fact that at a temperature of ~ 200°C, the ionic product of water is almost three orders of magnitude higher than at room temperature, accelerating hydrolysis. The hydrolysis of NBR and ABS in neutral media has not been studied. Overall, the potential of alkali-free hydrolysis lies in the absence of the formation of difficult-to-dispose wastewater, equipment corrosion, and polymer contamination with Na+ and K+ ions, which could affect further polymer recycling.
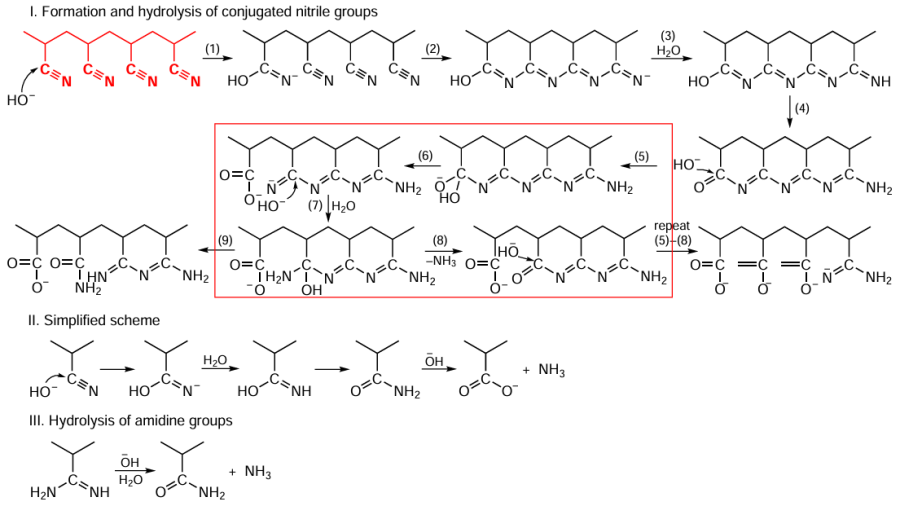
However, the greatest body of works in the field of PAN hydrolysis are devoted to alkaline hydrolysis, which is carried out at temperatures from 60 to 160°C and using solutions of NaOH[37][40][41] or Na2CO3.[45] Scheme 2 shows the main reactions occurring during hydrolysis in alkali solutions (NaOH, KOH).[46] Hydrolysis proceeds via the formation of amidine groups until almost complete conversion of nitrile groups is achieved. The main pathway for hydrolysis of chain units with a regular arrangement of nitrile groups (I) is the formation of polyconjugated rings (reaction 2) with their subsequent isomerization (reaction 3). Next, OH– attacks the carbonyl carbon atom (reaction 4) to form the corresponding intermediate (reaction 5), followed by opening of the ring (reaction 6), the amidine carbon atom group of which is attacked by OH– to form an amino group (reaction 7) with subsequent rearrangement and elimination of the NH3 molecule (reaction 8). Repetition of the cycle (5) – (8) results in a chain with a sequence of carboxyl and amidine groups. Amidine structures are stable even at hydrolysis temperatures above 75°C. Isomerization (9) and repeated cycle of the reactions (5) – (8) furnishes a chain containing amide, carboxyl, and amidine moieties. Pathway (II) is characteristic of chain units with single nitrile groups, as well as with an increase in the degree of conversion of nitrile groups during hydrolysis. Hydrolysis of amidine groups (III) leads to the formation of amide groups. In weakly alkaline solutions, for example, in a solution of Na2CO3 ,[45] hydrolysis does not afford amidine groups, following mechanism II.
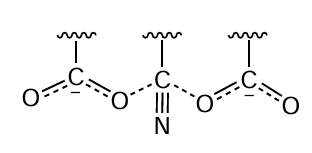
The polymer is not completely denitrogenated during hydrolysis (see Scheme 2); the chain preserves amino and amidine groups (if the latter are not hydrolyzed at temperatures above 75°C). According to a study,[45] during hydrolysis in Na2CO3 solution, some nitrile groups do not undergo transformation because they are deactivated by adjacent carboxyl moieties (Scheme 3). At a hydrolysis temperature of 105°C, the residual content of nitrile groups in hydrolyzed PAN is 38%.
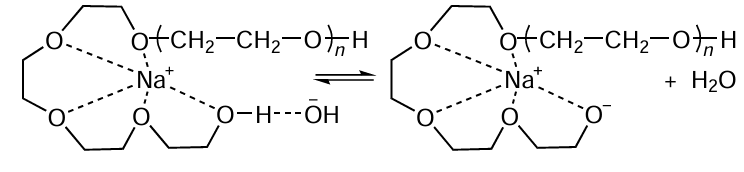
Research into the denitrogenation of nitrile-containing polymers is focused on selecting solvents, phase-transfer catalysts, and chelating agents that increase the accessibility of nitrile groups for attack by OH– ions. A series of studies[37][47][48] have shown that the degree of denitrogenation of spent ABS depends on the nature of the solvent and decreases in the order THF > dioxane > toluene. THF is characterized by greater solvent capacity for ABS, resulting in a looser polymer chain structure. Thus, the denitrogenation degree of ABS at a temperature of 120°C in THF is 44%, and at 160°C it is 93% with a hydrolysis time of 2 h in a NaOH solution.[48] The addition of chelating agents to the solution, e.g., ethylene glycol and polyethylene glycol (PEG) is of crucial importance to hydrolysis.[37][47][48] The authors explain the increased denitrogenation degree by the formation of chelates with a crown ether structure by PEG molecules, which leads to the effective solvation of the Na+ ion (Scheme 4) and increases its reactivity. Thus, in the absence of PEG, the denitrogenation degree of ABS is 36%, and with the addition of PEG-600 to the solution (5 mL per 1 g of ABS), it is 93%.
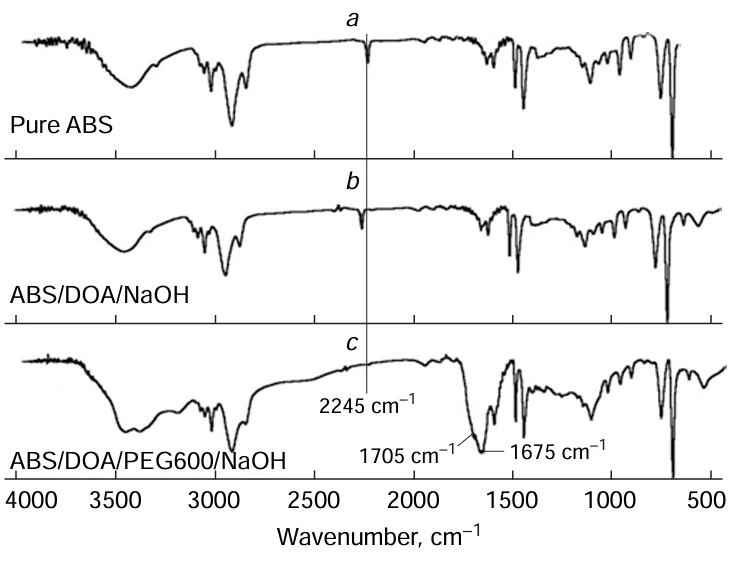
During hydrolysis, the reduction in nitrile group content is determined by IR (Fig. 3) and 13C NMR spectra. Sometimes, a slight decrease in the average molecular weight and the onset degradation temperature of ABS are observed, which are hallmarks of polymer chain degradation.
Hydrolysis as a method for debromination of ABS waste is not as effective as the denitrogenation method:[49] a debromination degree above 40% is achieved only at a temperature above 160°C, i.e. already under hydrothermal conditions.
An analysis of available research on ABS hydrolysis demonstrates the importance of hydrolysis in a neutral environment due to the absence of alkaline effluent formation and polymer contamination with alkali metal ions. Equally promising are studies examining the effect of stabilizers, plasticizers, and flame retardants contained in polymer waste on hydrolysis, as well as the study of NBR hydrolysis for denitrogenation.
4. Hydro- and solvothermal recycling
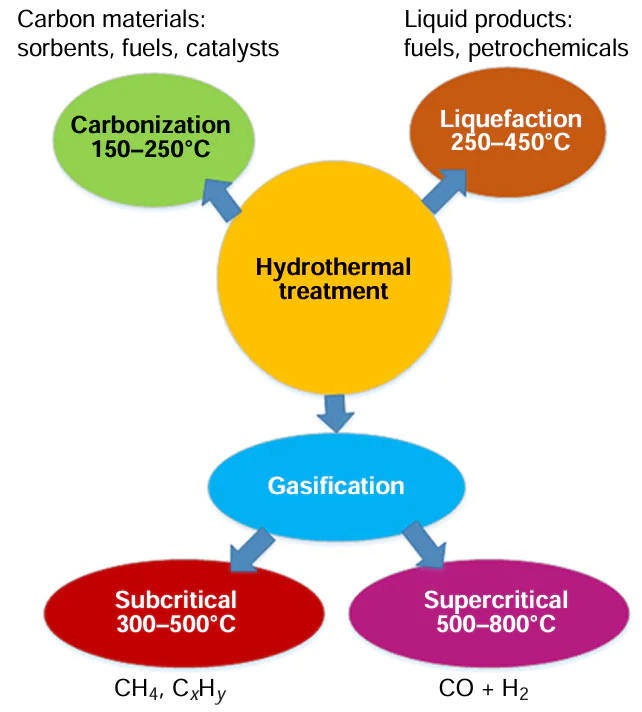
Hydrothermal recycling, in contrast to hydrolysis, involves the degradation of polymer chains during free-radical and ionic reactions and, in general, includes three processes that differ in temperature ranges and target products (Fig. 4).[1][50-52] In the subcritical region (below 374°C and 22.1 MPa), the ionic product of water increases with temperature, while the dielectric constant decreases. At temperatures close to the critical temperature, this causes water to exhibit properties similar to those of a non-polar solvent. Supercritical water is characterized by low viscosity, a high diffusion coefficient, a low dielectric constant, and good solvent capacity for many organic compounds, including non-polar ones.[1][52]
Hydrothermal liquefaction differs from pyrolysis in that it involves not only free-radical but also ionic reactions, which is important when recycling raw materials with a high content of heteroatoms (biomass, PET, ABS, PAN, polyurethanes, polycarbonate, PA, PVC, etc.), a lower gas yield, and a lower process temperature.[53] Therefore, hydrothermal processes open up wide possibilities for recycling plastic waste with a high content of functional groups, whereas polyolefins and PS under hydrothermal liquefaction conditions give a low liquid yield.[54]
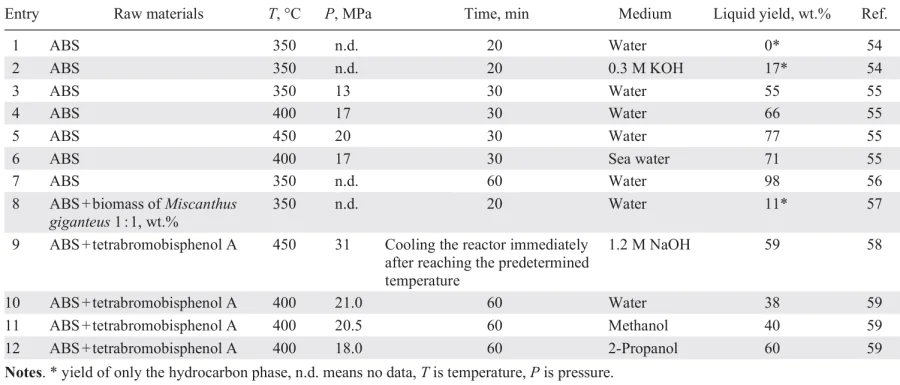
Not as many works have been devoted to the hydrothermal liquefaction of ABS (Table 1),[54-59] as to similar recycling of PET, polyurethane, PA, polycarbonate,[1][50][52][60-64] while the hydrothermal processing of BNK and PAN has not been studied.
Temperature has a significant effect on the yield of the liquid product. At temperatures below 300°C, virtually no liquid product is formed,[53][55][56] whereas at 350°C, the liquid yield increases significantly and continues to increase until reaching 450°C 55 (see Table 1, entries 3 – 5). The second important parameter of the process is its duration. At low temperatures (below 350°C), an increase in the reaction time promotes an increase in the liquid yield, whereas at high temperatures (above 350°C), the liquid yield increases significantly in the range of 30 – 60 min, then reaches a plateau.[52]
Hydrothermal liquefaction can be carried out both in pure water, and in aqueous solutions of alkalis, salts, or other solvents. When recycling pure ABS in the absence of alkali at a temperature of 350°C, no liquid hydrocarbon product was formed[54] (see Table 1, entry 2), while the yield of the aqueous phase was 9 wt.%. With the addition of KOH, the yield of the liquid hydrocarbon product increased to 18 wt.%, and the yield of the aqueous phase increased to 58 wt.%; with a significant portion of the liquid products of hydrothermal conversion being concentrated in the aqueous phase. Analyzing the data in Section 2, the role of alkali in the hydrothermal liquefaction of ABS waste is obvious: hydrolysis of nitrile groups delivers carboxyl groups, which, in turn, increase the hydrophilicity of the polymer chain and the solubility of its individual units in water (the chain becomes a polyelectrolyte), which favors its thermal degradation to afford low-molecular-weight compounds (Fig. 5). The resulting low-molecular-weight carboxylic acid salts are water-soluble and become part of the aqueous phase. This is why, in the hydrothermal recycling of nitrile-containing polymers, the liquid hydrocarbon product cannot be considered the primary target product, as many organic compounds are concentrated in the aqueous phase and, after extraction, can be used as valuable feedstock for the petrochemical industry. Replacing fresh water with sea water allows for a slight increase in the liquid yield from hydrothermal liquefaction of ABS waste (Table 1, entries 4 and 6),[55] while the proportion of polycyclic aromatic hydrocarbons in the liquid product increases and the H/C ratio decreases. This fact opens up prospects for studying the effect of salts on the course of hydrothermal liquefaction and makes it possible to use mineralized water, i.e. sea water, formation water separated during oil dehydration, etc., in this process.
The use of not only water (hydrothermal processing) but also organic solvents such as alcohols, phenols, alkanes, ketones, dioxane, and ethylene glycol has been widely studied for the recycling of plastic waste.[52][59][64] In the latter case, it is more correct to speak of solvothermal recycling. Since virtually all organic solvents have critical temperatures and pressures lower than those of water (e.g., 243°C, 6.37 MPa for ethanol), solvothermal recycling can be carried out under milder conditions. Furthermore, alcohols and phenols are more effective proton donors,[65] which increases feedstock conversion and suppresses polycondensation reactions. For efficient thermal degradation of solid particles/copolymer melt, its solubility in the solvent used is crucial, which depends, inter alia, on the solvent’s dielectric constant at the reaction temperature. Non-polar organic compounds are more soluble in solvents with low dielectric constants. However, alkaline hydrolysis of nitrile groups affords a polyelectrolyte containing COO– moieties in the chain (see Fig. 5). Non-polar groups in aqueous solutions interact with each other, causing the polymer chain to fold into a compact ball. Hydrophobic groups are located within the ball, while hydrophilic groups form the outer layer. The degree of extension of these groups increases with increasing solvent dielectric constant and, consequently, with intensification of electrolytic dissociation. In a less polar solvent, electrolytic dissociation is suppressed, polyelectrolyte swelling is eliminated, and the macromolecules curl.
When recycling ABS waste containing tetrabromobisphenol A in various supercritical solvents (see Table 1, entries 10 – 12),[59] a high liquid yield and a low gas yield were obtained using 2-propanol. Also, for this solvent, the absence of nitrogen-containing compounds in the liquid product was noted, in contrast to the product obtained in methanol. The conversions of ABS waste during recycling in supercritical methanol, 2-propanol and water at 400°C are 63%, 71% and 76%, respectively[59] (see Table 1, entries 10 – 12), while the dielectric constants of the said solvents at this temperature are equal to 9, 2 and 1.5, respectively. Obviously, heating of the raw material and the solvent at low temperatures promoted he rapid alkaline hydrolysis of nitrile groups, which occurs more effectively in the most polar solvent (in this case, water), in which a higher degree of extension of polymer chain units containing polar groups is achieved. In addition, with an increase in the temperature of the reaction mixture, destruction of the extended polar units occurs, and the destruction reactions of the hydrophobic fragments are facilitated by their good solubility in the solvent, which increases with decreasing dielectric constant. Accordingly, the highest conversion of acrylonitrile copolymers is achieved in water, which favors both the hydrolysis of nitrile groups and the thermal destruction of non-polar chain units under supercritical conditions due to good solubility in supercritical water.
Of special mention is that the use of HCOOH and formates as proton donors is possible, as shown by the example of solvothermal recycling of biomass and wastewater treatment plant sludge:[66-69] the content of organooxygen and organonitrogen compounds in the liquid product decreases, and the H/C ratio increases. The use of HCOOH together with palladium catalysts is proposed to promote hydrodeoxygenation and hydrodenitrogenation and suppress polycondensation reactions yielding the coke formation.[69] Solvothermal recycling of ABS waste in the presence of HCOOH has been studied only under gasification conditions at temperatures of 500 – 800°C;[70] an increase in feedstock conversion has been shown, due to the participation of H• radicals in initiating cracking reactions and suppressing coke formation. The liquid product obtained in the presence of HCOOH has a lower content of styrene and α-methylstyrene due to the hydrogenation of double bonds, and a lower content of polycyclic aromatic hydrocarbons (naphthalene, biphenyl, phenanthrene).
The hydrothermal recycling process, flexible in terms of feedstock composition, is a solution for the recycling of mixed waste stored in municipal solid waste (MSW) landfills. In this regard, the processing of polymer waste, including the spent ABS, in mixtures with various types of biomasses is widely studied.[1][50][57][71-73] Thus, during hydrothermal liquefaction of Miscanthus giganteus biomass with ABS[57] at a temperature of 350°C, the liquid yield was 35 wt.%, of which 11 wt.% was the hydrocarbon product and 24 wt.% was the aqueous phase (see Table 1, entry 8). Despite the fact that during co-processing with biomass, the yields of the liquid product from the liquefaction of ABS and biomass separately are not combined, the liquid product of the co-processing of ABS and biomass is characterized by a lower oxygen content and a higher heat of combustion than during the processing of pure biomass. This is due to the presence of compounds formed during the decomposition of ABS in the liquid product, i.e., ethylbenzene, styrene, and benzenebutanenitrile. Hydrothermal co-recycling of nitrogen-containing polymer waste and nitrogen-containing biomass (algae, wastewater treatment plant sludge, food waste)[74-76] will require hydrodenitrogenation of the resulting liquid product.
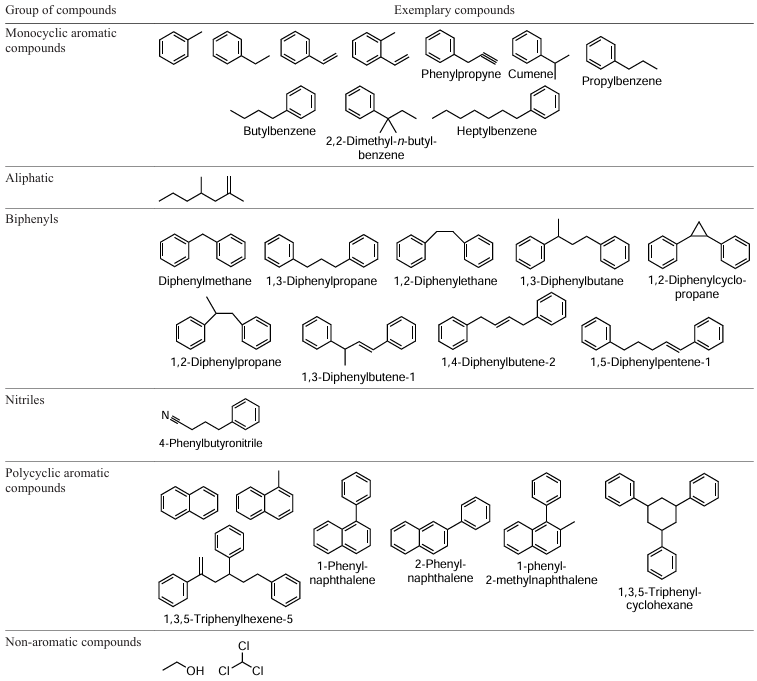
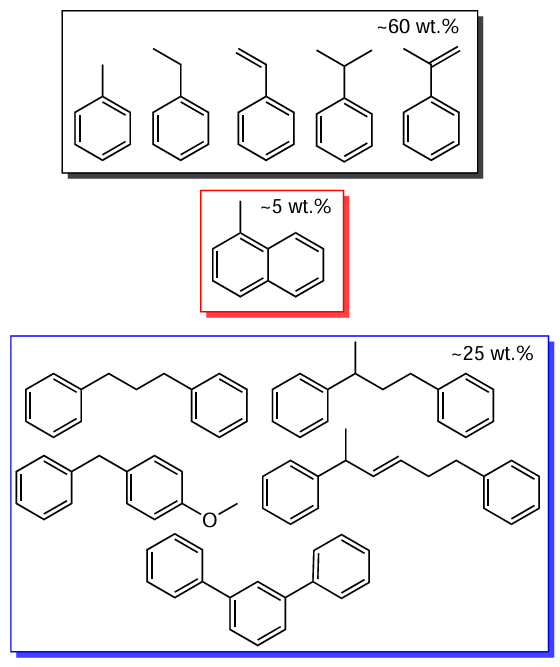
The liquid product of hydrothermal liquefaction of waste ABS comprises nitrile-containing aromatic compounds; hydrothermal liquefaction in an alkaline medium does not produce these compounds but gives NH2- and NH-substituted compounds.[54] The formation of compounds with the –C – N=C– group is also possible due to the addition of amines to butadiene units formed during depolymerization.[54][77] The main hydrocarbon components of the ABS liquefaction product are toluene, ethylbenzene, styrene, isopropylbenzene (1-methylethylbenzene), α-methylstyrene, the total proportion of which in the liquid product is about 60 wt.% (Scheme 5).[56] Heterodimers are also present (up to 25 wt.%) such as 1,3-diphenylpropane, 1,1'-(1-methyl-1,3-propanediyl)-bisbenzene, 1-benzyl-4-methoxybenzene and (E)-1,5-diphenylhex-3-ene, 1,1':3',1''-terphenyl. 1-Methylnaphthalene is present in an amount of about 5 wt.%.[53][55][56] Increasing the liquefaction temperature from 350°C to 450°C improves selectivity for ethylbenzene and polycyclic aromatic hydrocarbons and decreases selectivity for styrene and benzene dimers due to their thermal destruction.[55]
The ABS liquefaction product contains less than 1 wt.% of nitrogen, and increasing the process temperature from 400°C to 450°C helps to obtain a product that does not contain organo-nitrogen compounds.[55] During hydrothermal processing in an alkaline medium, the products contain no nitriles due to hydrolysis, but amines may be present. The main nitrogen-containing compound of the liquid product is benzenebutanenitrile; 2-phenyl-2-(propen-1-yl)pentene-4-nitrile, naphthalenecarbonitrile, and 2-[1-(4-cyano-1,2,3,4-tetrahydronaphthyl)]propanenitrile are also present.[57] When co-liquefied with biomass, the liquid product contains phenols, viz., 4-ethylphenol, 4-ethyl-2-methoxyphenol, and 2-methoxyphenol.[57]
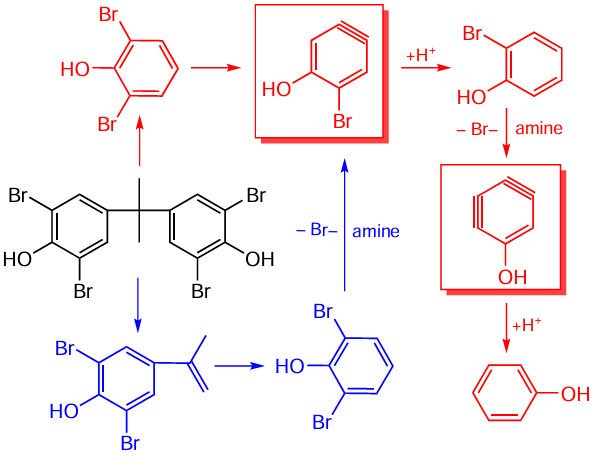
Hydrothermal conditions are very suitable for the removal of bromine atoms from bromine-containing flame retardants present in spent ABS.[49][58][59][78-81] Generally, to increase the degree of debromination, 1 – 3 M KOH,[60] K2CO3 ,[49][80] NaOH,[79] ammonia or amines are added to the water.[79] In an alkaline medium, a temperature of 260°C is sufficient to achieve a degree of debromination above 80%.[79][81] At this temperature, of the polymer chain does not degrade, and the resulting polymer can be sent to pyrolysis and also reused.[78] When using a K2CO3 solution at a temperature of 225°C, the degree of debromination above 90% is achieved with a feedstock/solution ratio of 1/2 and an anion ratio of CO32–/Br = 2/.[80] A comparison of the efficiency of various additives in the debromination of pure tetrabromobisphenol A at 260°C for 60 min gives the following order of increasing debromination degree: no additives (52%) < NaOH (60%) < NH3 solution (62%) < mono-, di-, and trimethylamines (94%).[79] Debromination in the presence of methylamine occurs already at 190°C (31%) and reaches 74% at 220°C. The main bromine-containing compounds of the product are 2,5-dibromophenol, 2-bromophenol, and 2,6-dibromo-4-(1-methylethenyl)phenol. Scheme 6 shows the reaction pathway for the stepwise debromination of tetrabromobisphenol A.[79]
Hydrothermal liquefaction of spent ABS containing tetrabromobisphenol A (the flame retardant), and Sb2O3 synergist was studied in an alkaline medium.[58]The liquid yield was 59 wt.% (see Table 1, entry 9), its main components were monocyclic aromatic hydrocarbons (toluene, ethylbenzene, propylbenzene, butylbenzene), indane, naphthalene and methylnaphthalenes, tetrahydronaphthalene, and biphenyl. No nitrogen-containing compounds were detected in the liquid product. The main products of flame retardant decomposition included phenol, 4-methylphenol, and acetophenone; bromophenols and unconverted tetrabromobisphenol A were present in trace amounts in the liquid product. In an alkaline medium, the released HBr is fixed to form NaBr and NH4Br. Antimony was also not detected in the liquid product. To increase the degree of debromination and reduce the reaction temperature, it was proposed to add Na2S to the solution in addition to the alkali.[78] Thus, hydrothermal treatment of WEEE provides the degree of debromination of 7% at 220°C in a NaOH solution (20 g L–1), and in a solution of NaOH (20 g L–1) + Na2S (50 g L–1) it is 90%; the antimony removal under the same conditions is 18% and 83%, respectively. Probably, the rise in the debromination degree upon the addition of Na2S to a NaOH solution is due to an increase in the total concentration of Na+ ions in the solution, and the degree of the antimony removal increases due to the formation of SbS33– ions.
In terms of debromination efficiency in the hydrothermal conversion of pure tetrabromobisphenol A at a temperature of 400°C without the addition of alkali, the solvents can be arranged in the following order: acetone (47%) < 2-propanol (64%), methanol (69%) < water (98%).[59] Debromination also occurs in the reaction medium in the absence of alkali; however, the resulting HBr dissolves in the liquid product, especially when using alcohols as a solvent rather than water,[59] due to the good solubility of HBr in alcohols and some polar compounds of the liquid product.
The addition of alkalis allows HBr to be bound into salts that are insoluble in the organic product.
As an alternative to alkalis, it has been proposed[81] to add PE and PP to high-impact polystyrene (HIPS) waste containing decabromodiphenyl oxide during debromination. With a 1 : 1 HIPS to PE waste ratio at 260°C, the debromination rate reaches 77%; for a 1 : 1 HIPS to PP mixture, the debromination rate is only 21%. Apparently, the small amount of H2 evolved during the thermal decomposition of polyolefins promotes the hydrodebromination of the flame retardant, although the conversion of this reaction in the absence of a catalyst is low. Nevertheless, this effect can be utilized in the recycling of polymer blends containing PS, ABS, and polyolefins.
Analyzing research in the field of hydrothermal recycling of ABS waste, it should be noted that this approach holds promise due to the almost complete denitrogenation of the polymer in the presence of alkali at liquefaction temperatures. Unfortunately, noteworthy is a lack of research aimed at studying the influence of the content of plasticizers, dyes, and stabilizers contained in polymers on the liquefaction process and product composition. One might assume that studying the effect of the solvent nature on the hydrolysis and solvothermal recycling of ABS is promising, since both the degree of extension of macromolecules due to the presence of charged units and the hydrolysis of nitrile groups, and the solubility of hydrophobic fragments in the solvent are important for the effective decomposition of polymer chains. One possible area for intensifying the hydrothermal recycling of ABS may be the use of proton donors, e.g., HCOOH, formates, and alcohols, to suppress coke formation. It is also possible to talk about the possibility of hydrothermal co-recycling of nitrogen-containing biomass and polymers with subsequent hydrodenitrogenation or hydrothermal recycling of a liquid product containing a residual amount of nitrogen.[82]
5. Gasification
Gasification of plastic waste of variable composition is a technologically and economically efficient method of processing it to produce methane and syngas. Numerous studies, including review articles,[83-87] have been devoted to this area, and their number is growing every year. In the vast majority of cases, gasification of polymer waste mixtures without preliminary separation is considered, which is a logical and economically viable solution for a technology that does not involve the production of monomers or petrochemical intermediates. A number of studies discusses co-gasification of polymer waste and biomass,[88-90] which is relevant for the recycling of real waste from the MSW landfills. Gasifying agents used included air,[84-90] water vapor (hydrothermal gasification),[91][92] and CO2.[93][94] Plasma gasification technology at temperatures above 1200°C has also been developed.[95][95]
Since a number of reviews have been devoted to the gasification of mixed plastic wastes,[83-87][96][97] we did not aim to cover this area in detail in this review. We will focus only on the studies considering gasification of pure ABS in order to assess the prospects of this approach to recycling its sorted waste.[92][93][95][98][99] During air gasification of polymers containing aromatic rings (ABS, PS), a high yield of char and a tar residue is observed, and in the case of gasification in a fluidized bed of sand or coal, carbon is deposited on the bed particles and the latter agglomerate.[98] Therefore, it is advisable to gasify these polymers in a mixture with waste from other polymers, such as polyolefins. Another solution is steam gasification,[92][99] which can significantly reduce the char yield. With an increase in the steam/feedstock ratio from 9/1 to 15/1, the carbon efficiency increases from 15% to 25% (and the hydrogen efficiency from 38% to 60%), calculated as the ratio of the carbon (hydrogen) content in the gaseous products to the carbon (hydrogen) content in the feedstock.[92]Plasma gasification in a CO2 stream at a temperature of about 1250°C makes it possible to obtain up to 92 wt.% synthesis gas from ABS copolymer (its composition includes 27 vol.% H2, 40 vol.% CO, 8 vol.% CH4, 22 vol.% CO2, 3 vol.% CxHy), up to 9 wt.% tar and up to 1 wt.% char.[93]
In any process of polymer waste gasification, tar is formed, its yield is maximum (up to 25 wt.%) in the case of hydrothermal gasification and minimum in plasma gasification; the tar yield during hydrothermal gasification passes through a maximum at a temperature of 700 – 800°C and then begins to decrease. The tar contains biphenyl, bibenzyl, terphenyl, naphthalene, methyl and ethyl naphthalenes, anthracene, 1,1'-binaphthyl, etc.[92][93][99]This product is of interest for further processing as a concentrate of aromatic hydrocarbons.
In summary, it can be concluded that gasification is impractical for processing sorted ABS, as such raw materials may be of interest as a source of ethylbenzene, styrene, amines, and carboxylic acids, which are products with a higher value than syngas. Gasification, as a non-selective process, is of great importance for processing mixtures of plastic waste of broad-based compositions.
6. Pyrolysis
6.1. Non-catalytic pyrolysis
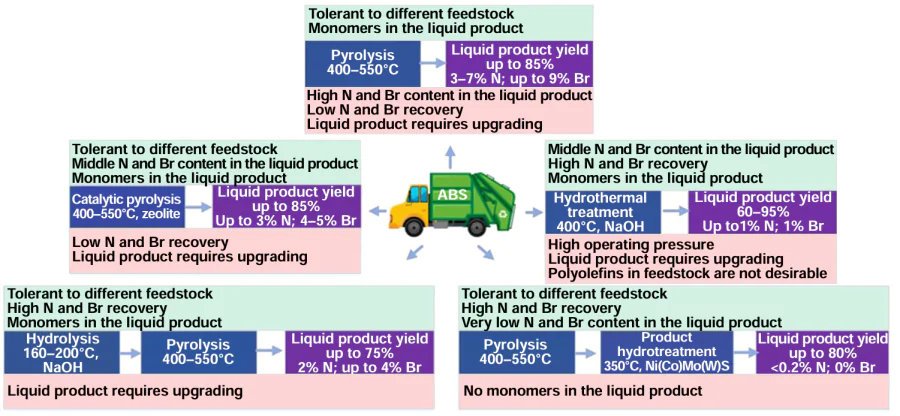
Among chemical methods of recycling plastic waste, pyrolysis ranks first in terms of the volume of waste processed and the number of companies engaged in the recycling of plastic waste based on this technology.[100] The main advantages of pyrolysis are the high liquid yield and the ability to recycle a wide range of plastic mixtures,[100-103] mixtures of plastic waste and biomass,[104][105] which cannot be converted into valuable products by other methods, such as solvolysis and hydrolysis. Pyrolysis compares favorably with hydrothermal recycling in that the process is carried out at low pressure. However, pyrolysis produces a mixture of products with a wide fractional composition, containing compounds of Cl, Br (if the raw materials contain PVC and flame retardants), N (during the pyrolysis of nitrogen-containing polymers), and O (during the pyrolysis of oxygen-containing polymers and polymers containing phthalate plasticizers). The presence of halogens in the pyrolysis feedstock creates a risk of corrosion of the pyrolysis unit and makes it impossible to use the liquid product as a fuel or feedstock for petrochemistry without dehalogenation.[102] A number of reviews have been devoted to the pyrolysis of plastics,[100-106] which examine the types of reactors (autoclave, fixed-bed, fluidized-bed catalyst, or adsorbent), the main types of catalysts (aluminosilicate, zeolite, Fe and Ca oxides, dolomite, etc.), and the possibility of combining pyrolysis with steam reforming to convert the products formed during pyrolysis into synthesis gas. Several works describe microwave-assisted pyrolysis.[106][107]
This review covers only the pyrolysis of acrylonitrile copolymers and their blends with other plastic wastes. We focused primarily on the composition of the resulting liquid product, primarily the redistribution of nitrogen- and bromine-containing compounds during pyrolysis, as this determines the possibility of obtaining high-added-value compounds from the pyrolysis product. The need for preliminary feedstock preparation and/or purification of the liquid product from heteroatoms largely determines trends in further research on the pyrolysis of heteroatom-containing polymers. We also analyze the mutual influence of mixed polymers on the yield and composition of pyrolysis products, and the potential for using catalysts to increase the yield of the liquid product and reduce its N and Br atom content.
The main options for reducing the N, Br, and Cl atom content in pyrolysis products are as follows:[10][12][13] [100-106]
— preliminary pyrolysis, which separates the bulk of the HCl and prevents it from further contact with the products;
— varying the rate and duration of raw material heating;
— using a combination of thermal pyrolysis and catalytic pyrolysis, where the pyrolysis products formed as a result of thermal destruction pass through a catalyst bed, where denitrogenation, dechlorination, and debromination occur.
According to TGA data,[108-111]ABS undergoes thermal decomposition in a single step in the temperature range of 230 – 500°C, with a weight loss peak at 330 – 440°C; as the heating rate increases, the weight loss peak shifts to a higher temperature region. Thermal decomposition of PAN occurs in two steps with maxima at temperatures of approximately 300°C and 580°C.[112] In the presence of a brominated flame retardant in the ABS copolymer, two decomposition peaks are observed on the TGA curves: the first peak in the range of 310 – 420°C corresponds to the decomposition of the flame retardant, which, in accordance with its purpose, must undergo thermal degradation before the polymer combustion occur,[109] the second peak corresponds to the decomposition of the polymer per se. In the case of thermal decomposition of plastic blends, their mutual influence manifests itself in a change in the onset degradation temperature: a less heat-resistant polymer lowers the onset degradation temperature of a more heat-resistant one. Thus, ABS lowers the onset degradation temperature of PVC, PA, and polycarbonate due to the participation of free radicals formed during the degradation of ABS chains in initiating cracking reactions of the second polymer.[110]
The onset degradation temperature of NBR[113-115] and ABS[108][111] is determined by the average molecular weight and structure, as well as the proportions of the constituent units. In general, an increase in pyrolysis temperature promotes the intensification of cracking reactions with the formation of low-molecular-weight products and an increase in the gas yield.[101][103] At temperatures up to 380°C, an increase in reaction time promotes an increase in the liquid yield, mainly due to overcoming the kinetic limitations of the thermal degradation reactions of the raw materials. However, at temperatures above 380°C, the effect of the reaction time is practically unnoticeable.[101]
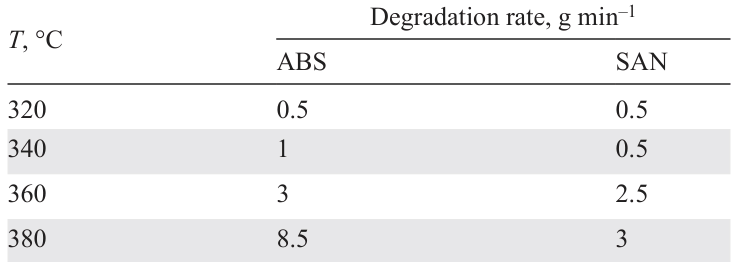
The rate of degradation of ABS at temperatures below 360°C is low and increases significantly with an increase in temperature to 380°C; the rate of decomposition of SAN increases five times with an increase in temperature from 340°C to 360°C (Table 2).[116]
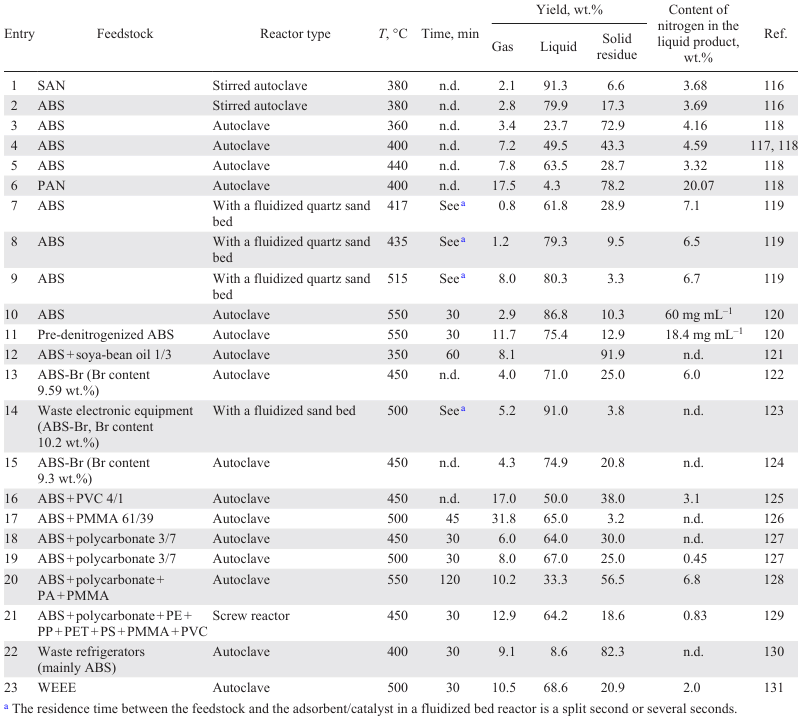
Let us analyze the effect of temperature on the yield of the products and the distribution of nitrogen-containing compounds in them for the pyrolysis of pure ABS without the addition of other polymers. Data on the parameters of non-catalytic pyrolysis[116-131] and the product yield are presented in Table 3. The liquid yield during pyrolysis increases with temperature, while the solid yield, on the contrary, decreases (see Table 3, entries 3 – 5).[116-118] The density of the liquid product increases with increasing pyrolysis temperature[118] because of the inclusion of higher-molecular-weight destruction products therein both due to the secondary polymerization reactions with increasing temperature and due to the deeper occurrence of thermal decomposition at elevated temperatures affording oligomeric units, which at low temperatures would remain unconverted and would become part of the solid residue.
When carrying out pyrolysis in a reactor with a fluidized bed of inert material,[119] the yield of products also increases with temperature: above 435°C, there is almost no increase in the liquid yield, but an increase in the gas yield is observed. Increasing the feed rate into the reactor from 2.6 to 4.5 g min–1 slightly (by 10 wt.%) increases the liquid yield and decreases in the yield of solid residue[119] due to a decrease in the residence time of cracking products in the reaction zone and a decrease in the proportion of side reactions of polymerization.
The liquid yield in the ABS pyrolysis is lower than during SAN pyrolysis (see Table 3, entries 1 and 2). Comparing the pyrolysis of PAN and ABS (Table 3, entries 4 and 6) shows that the main product of PAN pyrolysis is a solid residue, while the liquid product is distinguished by a very high nitrogen content (~ 20 wt.%)[118] due to its initially higher content in PAN (25.54 wt.%) than in ABS (6.65 wt.%). The absence of styrene and butadiene units in the PAN structure results in a low liquid yield and a high gas yield, since it is the products of thermal degradation of the butadiene – styrene units of the polymer chain that form the basis of the liquid product.
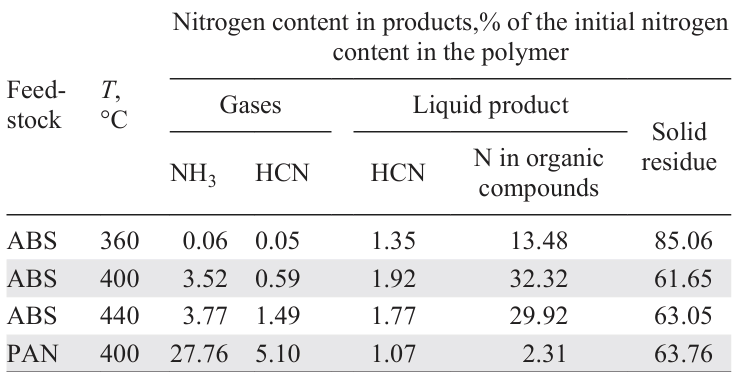
Table 4 shows the distribution of nitrogen in the products depending on the temperature of ABS and PAN pyrolysis.[118] The ABS pyrolysis in a batch reactor follows the following pattern: with an increase in temperature in the range of 360 – 400°C, the content of N-organic compounds in the liquid product increases, and then, with an increase in temperature, it decreases slightly due to the fact that at a low pyrolysis temperature, cyclization of acrylonitrile units affording nitrogen-containing fragments predominate, which at a higher temperature degrade to hydrocarbons. It was found[119] that the content of N atoms in the gaseous product decreases with an increase in the pyrolysis temperature in a fluidized bed in the range of 418 – 552°C, while it increases slightly in the liquid product due to an increase in the total conversion of the raw material and a decrease in the yield of solid residue.
The organic nitrogen content of the liquid product of PAN pyrolysis is lower than that of ABS pyrolysis, since cracking during ABS pyrolysis produces fragments containing acrylonitrile units in the molecule. PAN pyrolysis proceeds by a different plausible pathway:[118] the nitrile moieties are eliminated to form NH3 and HCN, which contain a significant proportion of the nitrogen from the starting polymer, and virtually no liquid product is formed.
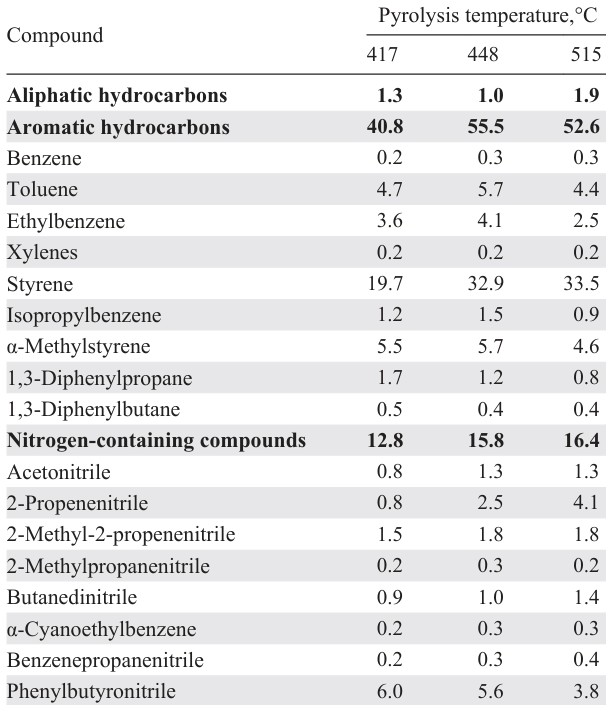
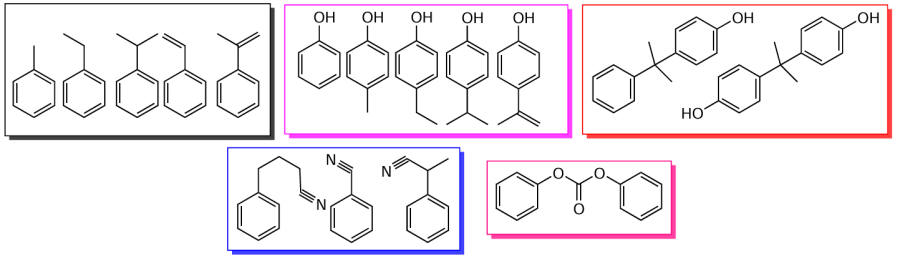
The main hydrocarbon components of the liquid ABS pyrolysis product comprise styrene, ethylbenzene, toluene, isopropylbenzene; among the nitrogen-containing compounds, the main one is phenylbutyronitrile (up to 6 wt.%); butanedinitrile, benzylnitrile, phenylacetonitrile, 1-naphthalenecarbonitrile, and methylquinolines are found in smaller quantities.[116-119] With an increase in the pyrolysis temperature, the content of aromatic hydrocarbons, including styrene, increases, and the content of phenylbutyronitrile decreases (Table 5).[119] The total content of nitrogen-containing compounds in the liquid product increases with the pyrolysis temperature, as discussed above for the total N content, while the content of phenylbutyronitrile, which is a product of the reaction between acrylonitrile and styrene,[118] decreases, probably due to the secondary cracking reactions of the resulting heterodimers, including phenylbutyronitrile, to give of low-molecular-weight nitriles (acetonitrile, propenenitriles), the content of which increases with temperature. Nitrogen-containing heterocycles were also detected in the ABS pyrolysis product carried out in an autoclave: methyl- and dimethylquinolines, N-benzylpyrrole, methylpyridines, imidazoles, and quinolineamines.
The content of ethylbenzene and isopropylbenzene decreases with increasing ABS pyrolysis temperature both in an autoclave[118] and in a fluidized bed reactor,[119] while in the autoclave an increase in the yield of diphenylpropane was observed with an increase in temperature from 360°C to 440°C.[118] The content of styrene increases with pyrolysis temperature in a fluidized bed[119] (see Table 5), but decreases in an autoclave reactor,[116] while the content of its dimers increases due to side reactions. The obtained patterns should help in optimizing pyrolysis conditions in order to increase the liquid yield, as well as to increase the content of styrene therein, the main target product of ABS pyrolysis.
Pyrolysis of SAN produces a liquid product with a higher styrene content (44.64 wt.% at 380°C) than during the pyrolysis of ABS (30.6 wt.%).[116] Pyrolysis of ABS delivers more alkylbenzenes (2 wt.% vs. 0.15 wt.% for SAN). The ABS pyrolysis product is distinguished by a higher content of phenylbutyronitrile, the total content of nitrogen-containing compounds is almost the same.
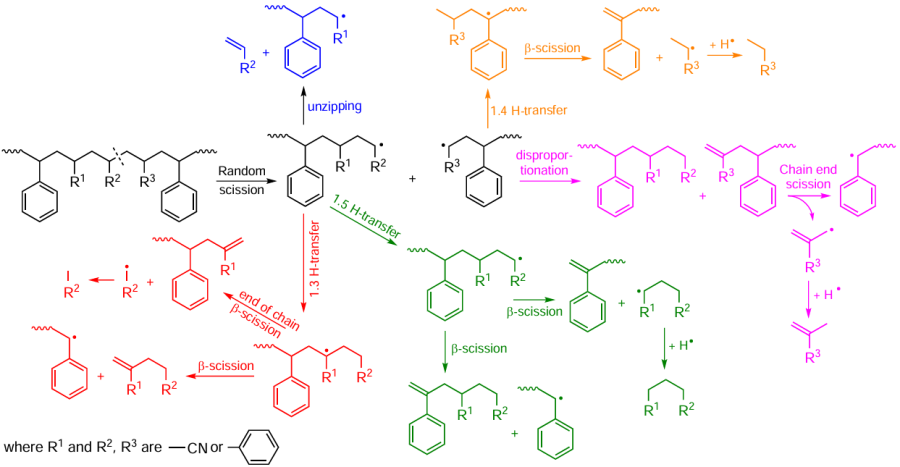
The mechanism of transformations occurring during non-catalytic pyrolysis of ABS is shown in Scheme 7.[132] Random scission of bonds in ABS molecules affords styrene–acrylonitrile macroradicals, which undergo further cracking, intramolecular hydrogen transfer, or disproportionation. Cracking delivers monomers (styrene, acrylonitrile). Hydrogen transfer followed by β-bond cleavage promotes the formation of heterodimers (3-hexenedinitrile, 3-butenylbenzene, benzylcarbonitrile, phenylbutyronitrile, 2-methylene-4-phenylbutyronitrile), heterotrimers (2-methylene-4-phenylheptanedinitrile, 4,6-dinitrile-hept-6-enenitrile) and oligomers, as well as toluene and ethylbenzene. Disproportionation results in a chain with unsaturated terminal bonds, which leads to the formation of macroradicals. Bond cleavage at the the chain termini affords methacrylonitrile and α-methylstyrene.
In the case of pre-denitrogenation of ABS by the hydrolysis method,[47][48] the initial degradation temperature and peak temperature decrease (according to TGA data).[120] During subsequent pyrolysis, the liquid yield is reduced, but the content of organonitrogen compounds in it is expected to decrease (see Table 3, entries 10 and 11). If during ABS pyrolysis 92.6% of the nitrogen content in the polymer are concentrated in the liquid product as N-organic compounds, then during the pyrolysis of denitrogenated ABS, this value decreases to 42.75%, and 43.42% of the N atoms move into the solid residue. The pyrolysis product of denitrogenated ABS contains less phenylbutyronitrile than untreated ABS (3.8 wt.% and 9.1 wt.%, respectively). The increase in the gas yield during the pyrolysis of hydrolyzed ABS is due to the presence of carboxyl groups, which are decarboxylated to release CO2.
Thus, pre-denitrification of ABS, SAN, and PAN prior to pyrolysis provides an opportunity to obtain a product with a low N content, and also eliminates the formation of HCN during pyrolysis and reduces the thermal stability of the polymer. Hydrolysis is particularly important in the recycling of plastics containing brominated flame retardants,[133][134] since reducing the Br content in liquid and gaseous pyrolysis products is a priority in WEEE recycling. This area encompasses a large body of studies, not only in relation to waste ABS, and certainly deserves a separate review.
In the presence of brominated flame retardants in the ABS composition, as already noted,[109][122]the onset degradation temperature of a copolymer decreases. In this case, the liquid yield is generally the same as during the pyrolysis of ABS without additives[122-124][135] (see Table 3, entries 13 – 15). The Br content in the liquid product depends on the pyrolysis mode: increasing the process time in the autoclave promotes the decomposition of the resulting intermediate organobromine compounds yielding hydrocarbons, therefore, the Br content in the liquid product can be about 0.7 – 0.8 wt.%, while the main part of the Br atoms is concentrated in the residue and heavy fraction of the liquid pyrolysis product (about 80% of the initial Br content in ABS).[122][124] During the pyrolysis of ABS in a fluidized bed, the short residence time of the raw materials and products in the high-temperature zone does not promote the degradation of bromine-containing semi-finished products, therefore, 97 – 99% of the Br atoms accumulate in the liquid product[123] (see Table 3, entry 14), which corresponds to the Br content in the liquid product of 8.9 wt.% with its content in the original polymer of 10.2 wt.%.
The major organobromine products of ABS pyrolysis containing tetrabromobisphenol A are 2,6-dibromophenol, 2-bromophenol, 3-bromophenol, dibromobisphenol A, bromobisphenol A, 2-bromo-4-(1-methylethyl)phenol, dibromomethylbenzenes, bromomethane, 1-bromobutane, 1-bromo-3-ethoxybenzene, and bromomethylbenzofuran.[123][124][135][136] A significant portion of the Br atoms in the liquid product is present in the form of SbBr3 ,[123] soluble in organic products, since Sb2O3 is a synergist and is always added to ABS and HIPS together with additives of brominated flame retardants in an amount of 3 – 4 wt.% Sb. The liquid product of ABS-Br pyrolysis also contains bisphenol A, phenol and alkylphenols, acetophenone, and benzofuran derivatives.[124][136] With an increase in pyrolysis temperature, the content of phenolic compounds decreases, while that of bromine-containing compounds remains virtually unchanged.[136]
Pyrolysis of ABS containing decabromodiphenyl ether produces polybrominated diphenyl ethers, diphenyl ether, bromophenols, phenol, bromobenzenes, and benzene.[136] An increase in temperature leads to a smaller amount of organobromine compounds in the products, since the decomposition and debromination of the starting compound are intensified.
In order to reduce the Br content in the liquid product, it was proposed[137] to carry out the pyrolysis of ABS-Br in the presence of NaOH or Ca(OH)2 . The pyrolysis product without additives contains 2-bromophenol in a concentration of 134 mg L–1, 2,4-dibromophenol (125 mg L–1), and 3-bromophenol (~ 40 mg L–1). During pyrolysis in the presence of Ca(OH)2 at a ratio of ABS-Br/Ca(OH)2 = 5/1, the content of these compounds decreases to 40, 55, and 15 mg L–1, respectively, and during pyrolysis with the addition of NaOH at a ratio of ABS-Br/NaOH = 10/1, the product contains only 2-bromophenol in a concentration of 12 mg L–1. However, despite the high debromination degree, pyrolysis with alkali additives is not without its drawbacks, including the formation of salts that become part of the solid pyrolysis residue, which makes the regeneration of alkali and bromine-containing compounds challenging.
In recent years, research has been conducted into the creation of halogen-free flame retardants. In particular, phosphorus-containing flame retardants for ABS copolymer, aluminum hypophosphite and aluminum alkyl phosphinates, for which red phosphorus acts as a synergist, are being tested.[138] Unfortunately, studies on the pyrolysis of acrylonitrile copolymers with phosphorus-containing flame retardants are sporadic[127] because these flame retardants are only just beginning to be used in the production of polymer products, and such products have not yet been recycled. However, pyrolysis of a mixture of ABS and polycarbonate with an oligophosphate flame retardant at a temperature of 450 – 550°C produces the liquid product that does not contain organophosphorus compounds.[127] This fact gives hope that the future switch to phosphorus-containing flame retardants will simplify the chemical recycling or reuse of spent ABS. Nevertheless, the task of chemical recycling of plastic waste with brominated flame retardants, as well as the problem of debromination of pyrolysis products and the creation of catalytic systems that allow debromination to be carried out simultaneously with pyrolysis, remains very urgent.
ABS, like PVC, HIPS, polycarbonate, and PMMA, is widely used in the production of electronics and household appliances. Real WEEE is a mixture of these polymers with added flame retardants, stabilizers, plasticizers, and dyes. Separating this mixture before pyrolysis is often impossible, so studies of the pyrolysis of real WEEE[128-131][139] or mixtures of the polymers that make up WEEE are of great interest.[125-127]
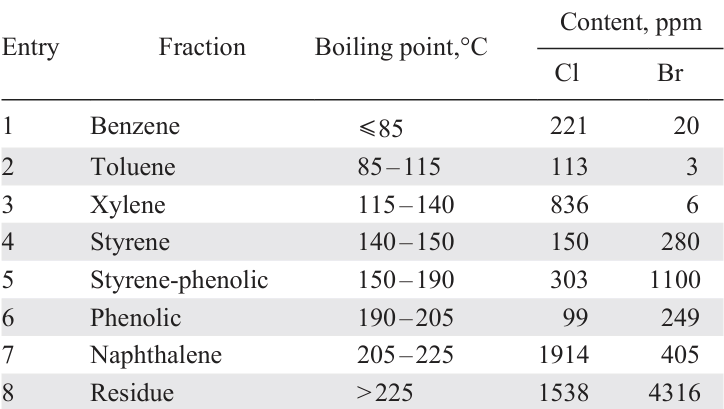
PVC is the least desirable component for pyrolysis together with ABS, since the addition of PVC decreases the liquid yield (from 72% for ABS to 50% for the ABS + PVC 4/1 mixture, see Table 3, entry 16) and contamination of the product with organochlorine compounds (Cl content is 0.42 wt.%).[125] At the same time, the N content in the product decreases slightly due to the interaction of nitrogen-containing compounds with HCl and moving of the resulting salts to the solid residue. The reaction of acrylonitrile formed during pyrolysis with HCl lowers the content of C11 – C14 compounds, products of di- and trimerization of acrylonitrile and styrene,[125] in the liquid product, while the styrene content increases. The main chlorinated compounds in the liquid product include 1-chlorobutane, 1-chloroethylbenzene, 2-chloro-1,4-dimethylbenzene, 3-chloropropanenitrile, and 2-chlorophenol (when the ABS copolymer contains tetrabromobisphenol A).
PMMA can be considered to be a desirable component in the pyrolysis of ABS,[126] since its degradation begins at a lower temperature than the degradation temperature of ABS, and the resulting free radicals initiate the cracking of ABS macromolecules. Despite the moderate yield of the liquid product (see Table 3, entry 17) compared to that of the liquid product of the pyrolysis of pure ABS, the thermal degradation of this mixture is attractive in terms of using the liquid product as a motor fuel, which is distinguished by a high octane number (110) due to the presence of oxygen-containing compounds —products of the degradation of PMMA and the dimerization of methacrylate units (M) with units of styrene (C), butadiene (B) and acrylonitrile (A).[126] The liquid product contains methyl methacrylate, ethyl heptanoate, methyl 4-phenyl-2-methylbutanoate (heterodimer SM), other heterodimers of SM, heterotrimers of ASM of various compositions.
During the pyrolysis of mixtures of ABS and polycarbonate (see Table 3, entries 18 and 19), the liquid yield is comparable to that for the pyrolysis of ABS and PMMA mixtures.[127] Scheme 8 shows the main compounds of the liquid product of the pyrolysis of a mixture of ABS and polycarbonate.[127]
The main product of polycarbonate decomposition is bisphenol A. Its content in the liquid product reaches 30 wt.% and decreases with increasing pyrolysis temperature; the phenol content in the product is about 13 wt.% and is little affected by temperature. Pyrolysis of a mixture containing ABS, polycarbonate, PMMA, and PA (see Table 2, entry 20) gives a liquid product containing mainly 2,4,6-trimethylphenol, 4-chloro-3-n-hexyltetrahydropyran, dimethylnaphthol, with an oxygen atom content of 8.2 wt.%.[128] The possibility of pre-separation of polycarbonate from the WEEE mixture by extraction with N-methylpyrrolidone is reported.[128]
During the pyrolysis of spent refrigeration equipment consisting of a mixture of polymers (ABS + polycarbonate + PE + PP + PET + PS + PMMA + PVC) (Table 6), a liquid product containing 0.05 wt.% Cl and 0.02 wt.% Br is formed in a screw reactor (see Table 3, entry 21).[129] Its main components are styrene (28.1 wt.%), benzene, toluene and xylenes (13.4 wt.%), α-methylstyrene (4.7 wt.%). Among the acyclic hydrocarbons, 2,4-dimethyl-heptene-1, a characteristic product of the PP pyrolysis, predominates. The characteristic degradation products of PMMA and polycarbonate — methyl methacrylate and bisphenol A — were not detected in the product, likely due to the low content of the starting polymers in the mixture and also because their peaks in the chromatogram overlap with those of other polar compounds.[129] Indenes, naphthalenes, diphenyls, and tricyclic aromatic hydrocarbons also present. The presence of tricyclic aromatic hydrocarbons in the liquid product, which are products of side polycondensation reactions, was also observed during the pyrolysis of spent printers: acenaphthylene, fluorene, and anthracene were identified.[139]
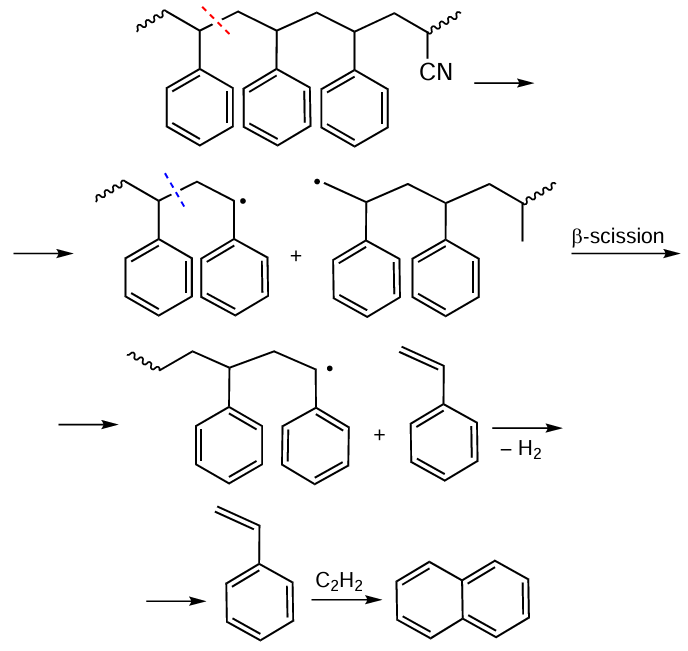
Scheme 9 illustrates a possible mechanism for the formation of naphthalene during SAN pyrolysis. The presence of polycyclic aromatic hydrocarbons (PAHs) in the liquid pyrolysis product creates additional difficulties for its use, since their isolation is impractical at such a low concentration (≤ 3 wt.%), but the use of the liquid product as a fuel without prior dearomatization becomes impossible. Dearomatization is possible only after denitrogenation and dehalogenation, since PAH hydrogenation catalysts, both based on platinum group metals and sulfide-based ones, are sensitive to impurities containing nitrogen and halogen atoms.
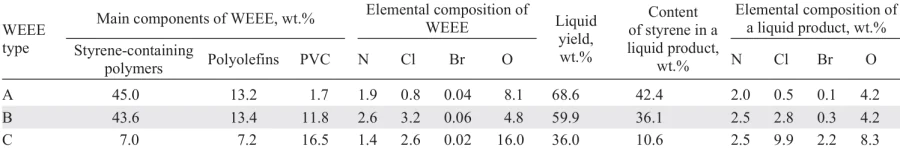
A comparison of the pyrolysis results for three different WEEE samples 131 (Table 7) shows that the liquid yield is highest in the case of pyrolysis of a plastic blend enriched with PS, polyolefins, and ABS. At a high PVC content, the liquid yield is low, both due to the low liquid yield of the pure PVC pyrolysis and due to the adsorption of HCl on the surface of coke particles with the formation of acid sites responsible for the occurrence of side reactions of polycondensation of unsaturated compounds affording high-molecular-weight tarry products.[140] The high content of styrene copolymers in the raw material subjected to pyrolysis ensures a high styrene content in the product (for comparison, in the pyrolysis product of mixtures of A, B and C it is 42.4, 36.1 and 10.6 wt.%, respectively) and its easy isolation, for example, by distillation or extraction methods.[131] The pyrolysis product of mixture C contains 7.6 wt.% caprolactam, the major product of the thermal degradation of PA.
The pyrolysis product of the C mixture is characterized by a very high content of Cl, Br, and O atoms (see Table 7). Its use as a raw material for further recycling or as a fuel is impossible without refining, and the low styrene content makes its isolation economically inexpedient. Therefore, for such waste plastic blends characterized by a high content of PVC, PA, it is advisable to either carry out pre-separation, e.g., by extraction, to separate oxygen-containing polymers,[128]hydrothermal recycling in order to reduce the Cl content,[141][142] or carry out pyrolysis/carbonization aimed at obtaining a solid product, hydrocarbon gases or H2.[143][144]
6.2. Catalytic pyrolysis
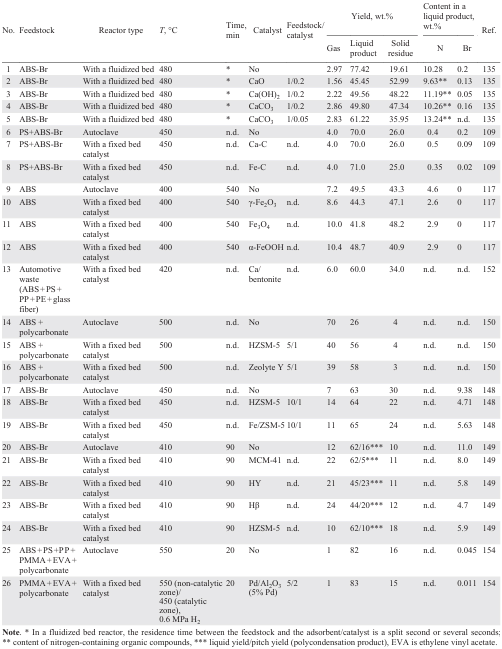
Partial debromination, denitrogenation, and dechlorination of the liquid product are also possible using catalysts employed in the pyrolysis of various feedstocks. The main research in the field of catalytic pyrolysis of WEEE, including ABS-Br, is focused specifically on debromination and denitrogenation of the liquid product. The catalysts used include CaO, Ca(OH)2 , CaCO3;[109][135][145] iron oxides and oxyhydroxides;[109][117][146][147] zeolites,[124][131][148-150] including those with Fe additives;[148] industrial zeolite-containing catalysts for cracking petroleum fractions;[114] clays and aluminosilicates;[151][152] Al2O3;[153][154] catalysts improving the selectivity of pyrolysis for hydrogen include Ni/SiO2, Co/SiO2 .[144]Table 8 summarizes the data on the catalysts used and the pyrolysis results.
Calcium and carbon composites (obtained by impregnating polymer resins with solutions of inorganic Ca salts followed by calcination),[109] CaO, Ca(OH)2 or CaCO3 ,[135] are used as Ca-based catalysts, but only for the purpose of reducing the Br content in the liquid pyrolysis product, while its yield does not increase or even decreases compared to the non-catalytic process.[135] Thus, thermal degradation of ABS-Br at a temperature of 480°C in a fluidized bed reactor provides 77.4 wt.% yield of the liquid product (see Table 8, entry 1). Using catalytic pyrolysis, the yield is 45.4, 49.6, 49.8 wt.% for catalysts CaO, Ca(OH)2 , CaCO3 , respectively (see Table 8, entries 2 – 4), with the liquid yield decreasing with an increase in the catalyst/raw material ratio (see Table 8, entries 4 and 5).[135] With an increase in the catalyst loading, the content of styrene, toluene, xylenes, and ethylbenzene in the liquid product decreases, while the content of organonitrogen compounds remains practically unchanged or even increases (for thermal degradation, this value is 10.28 wt.%), which is consistent with the data of study[109] for the Ca – C catalyst.¶¶ Thus, the N content in the liquid product of non-catalytic pyrolysis of a mixture of PS and ABS-Br is 0.4 wt.%, while during pyrolysis in the presence of 0.5 wt.% of the Ca – C catalyst (see Table 8, entry 7).[109] The minimum content of organobromine compounds is achieved in the pyrolysis product in the presence of Ca(OH)2. In contrast, CaCO3 is practically not involved in debromination. The used of the Ca – C catalyst reduces the content of Br atoms in the liquid product by 2 times compared to non-catalytic pyrolysis.
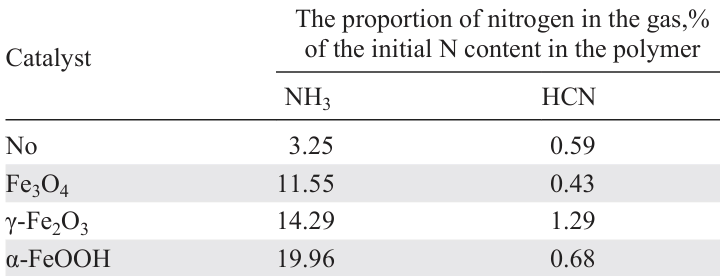
Catalysts based on iron oxides, hydroxides, and carbon-containing composites are somewhat more effective in reducing of both N and Br contents in pyrolysis products.[109][117][146][147] A comparison of performances of Fe – C and Ca – C catalysts in the pyrolysis of a mixture of PS and ABS-Br (see Table 8, entries 6 – 8) shows that the iron-containing catalyst provides low degree of denitrogenation and a significant reduction in the Br content in comparison with the thermal degradation and catalysis using the Ca – C composite; however, the liquid yield value does not differ from that for the thermal process. A comparison of catalysts based on iron oxides and iron oxyhydroxide shows (see Table 8, entries 9 – 12)[117][146][147][151] that, along with some decrease in the liquid yield, these catalysts provide effective denitrogenation; the minimum loss in the liquid yield is achieved in the presence of α-FeOOH at the same level of denitrogenation. During the catalytic process, the majority of N atoms move to the solid residue, and the yield of NH3 also increases by 3 – 6 times. It is important to note the activity of α-FeOOH in conversion of heavy nitrogen-containing C14 – C21 compounds to light C5 – C6 nitriles:[147][151] the content of light aliphatic nitriles in the product increases in the series of catalysts γ-Fe2O3 < Fe3O4 – C < α-FeOOH. This fact suggests the economic viability of separating light nitriles from the product by distillation and using them in petrochemical synthesis. The liquid product remaining after distillation is characterized by a low N content and can be used to separate toluene, xylenes, styrene, and ethylbenzene. However, an unstable phase composition of the α-FeOOH catalyst during pyrolysis has been reported,[146] since at a temperature of 400°C it is converted to Fe3O4.
Due to the ineffectiveness of Ca and Fe compounds in increasing the yield of the liquid pyrolysis product and their low activity in denitrogenation, zeolite-containing catalysts and aluminosilicates have attracted considerable attention. For example, during pyrolysis of spent monitors (PMMA + ABS) at 400°C over an industrial zeolite-containing cracking catalyst, the liquid yield increases from 8.6 (thermal pyrolysis) to 55.7 wt.%.[130] During pyrolysis of the ABS + polycarbonate mixture (see Table 8, entries 14 – 16), zeolites Y and ZSM-5 also provide a significant increase in the liquid yield,[150] while the content of monocyclic aromatic hydrocarbons in it increases significantly (styrene: from 2.47 wt.% for the thermal process to 36.37 wt.% for zeolite Y and 46.72 wt.% for ZSM-5; ethylbenzene: from 1.18 wt.% to 10.2 wt.% and 8.57 wt.%, respectively); the content of phenols and organobromine compounds also decreases noticeably (from 2.12 wt.% to 0.53 wt.% and 0.53 wt.%, respectively).
Fe/ZSM-5 and Fe/Y catalysts show higher debromination activity than the HZSM-5 and HY zeolites, most likely due to the adsorption of bromine compounds yielding FeBr3.[150] These data are contradicted by the results of work,[148] which indicate less efficient or similar debromination over Fe/ZSM-5 than HZSM-5. The HZSM-5 catalyst is characterized by a high surface area and acidity, these parameters decrease upon addition of Fe. The reduced cracking performance of the catalyst results in low activity in the conversion of bromine-containing compounds. Increasing the Fe content in the zeolite can provide a higher debromination degree, which occurs due to irreversible adsorption affording FeBr3 , making its regeneration impossible; i.e., the catalyst in this case functions as an adsorbent. Therefore, a higher debromination degree on iron-containing zeolites is observed for a short time, until the catalyst is deactivated. The replacement of some protons in the zeolite structure with Fe atoms leads to an expected decrease in its activity in cracking reactions and a decrease in the liquid yield.[150] Therefore, in general, the use of zeolites in the protonated form is preferable, and a reduction in the Br content in the liquid product can be achieved in a two-step pyrolysis process, when the gas formed in the first reactor (over a zeolite-containing catalyst) at the reaction temperature enters the second reactor containing a different catalyst (CoMo/Al2O3, NiMo/Al2O3, Ca – C).[124] In this case, a high liquid yield can be achieved with a low content of N (0.9 – 1 wt.%) and Br atoms (0.05 – 0.1 wt.%).
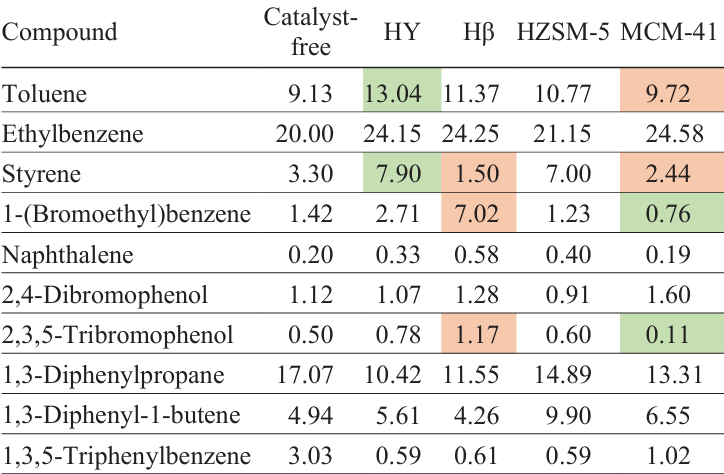
A comparison of the activity of zeolites HY, Hβ, HZSM-5 and aluminosilicate MCM-41 (Ref. 149) in the ABS-Br pyrolysis shows that aluminosilicate has virtually no effect on the yield of the liquid product and its composition; the process is similar to non-catalytic pyrolysis due to the low acidity of MCM-41. The use of zeolites HY and Hβ as catalysts decreases the liquid yield due to the side polycondensation reactions of monomers, heterodimers and heterotrimers formed during pyrolysis (see Table 8, entries 20 – 24), which is consistent with the data on the increase in the pitch yield. Pyrolysis over HZSM-5 zeolite does not decreases the liquid yield due to its smaller surface area (287.3 m2 g–1) than that of HY (671.0 m2 g–1) and Hβ (525.0 m2 g–1). Among the above zeolites, HZSM-5 has the highest acidity. Zeolites HY and Hβ provide more efficient debromination of the liquid product than MCM-41 and HZSM-5, apparently due to the intense polycondensation of bromine-containing compounds occurring on HY and Hβ, resulting in more bromine compounds being concentrated in the pitch.
Table 9 presents data on the content of the main compounds in the liquid product during pyrolysis over various zeolites and aluminosilicate MCM-41.[149] In the presence of zeolites, the cracking of macromolecules is intensified, therefore the yield of monocyclic aromatic hydrocarbons increases, and that of heterodimers and heterotrimers decreases. Zeolite catalysts, however, do not provide a high debromination degree of the product; the concentration of bromobenzenes and bromophenols is higher than for the product of thermal pyrolysis. The decrease in the total Br content in the liquid product using zeolite catalysis, as already noted, is due to the redistribution of Br atoms during polycondensation reactions and an increase in the proportion of high-molecular-weight bromine-containing compounds concentrated in the pitch. An increased amount of bromobenzenes and bromophenols in the liquid product under zeolite catalysis may also be due to the intensification of reactions of HBr with unsaturated thermal degradation products.[149]
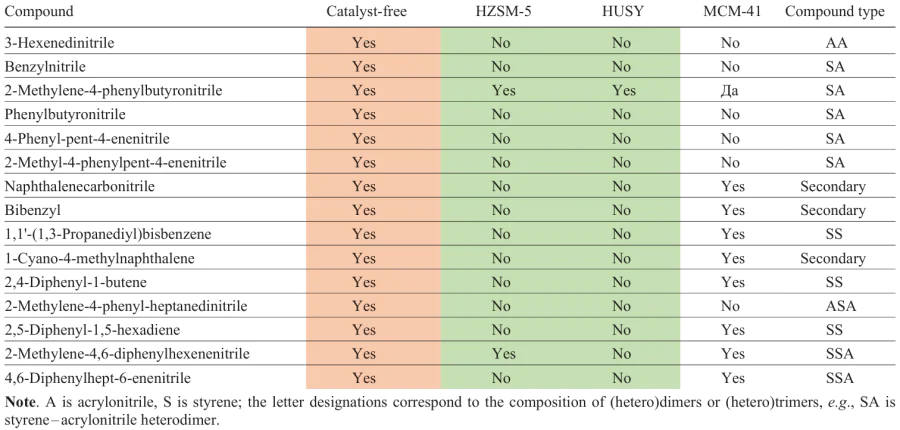
A comparative analysis of the ABS pyrolysis products in the absence of a catalyst, over aluminosilicate MCM-41 and over zeolite catalysts HZSM-5, HUSY[132] shows the absence of phenylbutyronitrile, benzylnitrile, 4-phenyl-pent-4-enenitrile, 2-naphthalenecarbonitrile and other nitrogen-containing heterodimers and heterotrimers in the composition of the catalytic pyrolysis products (Table 10). A catalyst-free pyrolysis yields a large number of heterodimers and heterotrimers, e.g., AA, SA, ASA, SSA (see Table 10), which are the main source of N atoms in the product, and in the presence of zeolites, heterodimers and heterotrimers undergo cracking to give NH3, acetonitrile, and acrylonitrile.[155] Similar to the data of work[149], the pyrolysis product obtained using MSM-41 (Ref. 132) differs little in composition from the product of non-catalytic pyrolysis and contains a significant amount of heterodimers and heterotrimers. The HUSY catalyst, characterized by a high concentration of strong Brønsted acid sites (98% of the total number of Brønsted sites, total acidity of 1153 μmol g–1 by TPD-NH3), is more active in the cracking reactions of nitrogen-containing heterodimers and heterotrimers than HZSM-5 with a lower concentration of strong Brønsted acid sites (80%, total acidity of 489 μmol g–1 by TPD-NH3).
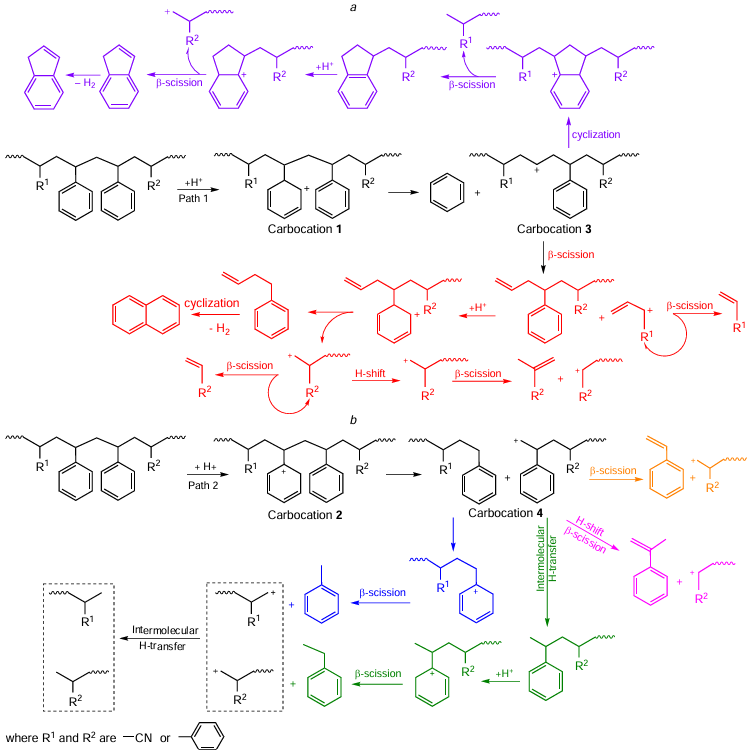
The mechanism of pyrolysis in the presence of a catalyst (Scheme 10) differs fundamentally from that of thermal pyrolysis (see Scheme 7).[132] Protons of the Brønsted acid centers attack the benzene ring to form carbocations 1 or 2. Carbocation 1 is involved in the formation of a benzene molecule. Carbocation 2 undergoes bond cleavage in the middle of the chain to form a heterotrimer of SAN and carbocation 4 with a positive charge on the terminal carbon atom, from which styrene is formed via the subsequent scission of the β-bond, or α-methylstyrene is formed upon hydrogen transfer followed by β-cleavage. With a sufficient number of Brønsted acid sites, heterotrimers and heterodimers serve as substrates in cracking reactions yielding monocyclic aromatic hydrocarbons. Indane and indene are formed from carbocation 3 as a result of cyclization followed by β-cleavage and dehydrogenation. Carbocation 3 also acts as a source of a naphthalene core via the intermediate 3-butenylbenzene, which is converted to naphthalene through dehydrogenation and cyclization.
The formation of a particular carbocation is determined by the acidity of the zeolite. Zeolite HUSY favors intramolecular hydrogen transfer, therefore, the yield of ethylbenzene in the presence of this catalyst is maximum, and that of styrene is minimum: carbocation 4 undergoes intramolecular hydrogen transfer to give ethylbenzene.[155] Moreover, zeolite HUSY inhibits the formation of indane and indene: carbocation 3, probably as a result of intramolecular hydrogen transfer, is converted into a macromolecule without cyclization. Zeolite HUSY, being involved in hydrogen transfer reactions, promotes the formation of NH3 and light nitriles.
Analysis of 155 HUSY zeolite spent in the pyrolysis of nitrogen-containing polymers shows that the N content in the catalyst increases from 0.4 wt.% for the fresh catalyst to 1 wt.% for the spent one but during regeneration at 500°C it decreases to 0.5 wt.%. As a result of deactivation, the total pore volume decreases from 0.438 to 0.088 cm3 g–1 and the micropore area decreases from 1016 to 18 m2 g–1. These indicators are restored during regeneration (0.435 cm3 g–1 and 908 m2 g–1), which provides the possibility of reusing the catalyst.
In general, zeolite-containing catalysts with high Brønsted acidity, such as HUSY and HY, are promising catalysts for combining thermal and catalytic pyrolysis, in which the thermal pyrolysis products pass through a catalyst bed in which cracking, debromination, and denitrogenation of oligomers take place.[156][157]
In conclusion of the discussion of catalytic pyrolysis, mention should be made of hydropyrolysis carried out under H2 pressure in the presence of hydrogenation catalysts. Thus, for recycling a mixture of plastic wastes containing ABS, PS, PP, PMMA, etc., it was proposed[154] to carry out a combination of thermal and catalytic hydropyrolysis at different temperatures in non-catalytic and catalytic reaction zones over a Pd/Al2O3 catalyst (see Table 8, entries 25 and 26). The use of a catalyst has virtually no effect on the product yield, but it does contribute to a significant reduction in the content of chlorine, bromine, nitrogen, and oxygen-containing organic compounds in the liquid product compared to the product of non-catalytic pyrolysis. However, the expected hydrogenation of styrene and α-methylstyrene occurs simultaneously; these compounds are not present in the product of catalytic hydropyrolysis, which is undesirable for the subsequent use of the liquid product for the isolation of these monomers. The palladium catalyst is probably rapidly deactivated under the conditions of processing such ‘complex’ raw materials with a high content of N, Cl, and Br atoms, which is generally characteristic of hydrodehalogenation over noble metals.[158] However, the concept of hydropyrolysis, including the sequential pyrolysis – hydropyrolysis process, may be attractive for recycling plastic waste into fuel using inexpensive, stable catalysts.
Summing up the analysis of research into pyrolysis of acrylonitrile copolymers, the main problem characteristic of the pyrolysis of WEEE should be noted — the high content of N and Br atoms (up to 10 wt.%) in the liquid product, which requires subsequent refining, the implementation of which in the processes of hydrodenitrogenation and hydrodehalogenation is undesirable, since it will lead to the loss of valuable monomers, viz., styrene and α-methylstyrene. Preliminary separation of styrene prior to hydrotreatment is difficult due to intense corrosion of equipment by bromine and chlorine compounds. Therefore, it may be preferable to prepare plastic waste for pyrolysis (by alkaline cleaning, hydrolysis, hydrothermal treatment, etc.) to obtain polymer feedstock with low halogen and N content, as well as to implement two-step pyrolysis (non-catalytic and catalytic steps) to reduce the formation of organic N, Br, and Cl-containing compounds.
¶¶ A composite obtained by impregnating a polymer resin with a CaCO3 suspension followed by calcination.
6.3. Use of a solid residue from pyrolysis and ammonia recovery
The yield of solid pyrolysis residue depends on the reactor type and the ash content of the feedstock and can reach 80 wt.% (see Table 3 and Table 8). During the pyrolysis of printed circuit boards, metal – polymer composites, and waste from household appliances and electronics, a solid residue is formed that is a valuable raw material for the extraction of metals such as Cu, Zn, Sb, Al, Fe, Cr, Co, Ni, Pb, Mn,[129][150] the total concentration of which can exceed 2.4 wt.%.[131] However, the use of solid pyrolysis residue as fuel or coke in metallurgy is difficult due to its high ash content and the presence of halogen atoms.[159] Solid pyrolysis residue also contains bromine atoms in the form of organically bound bromine and metal bromides, which opens up opportunities for bromine recovery. During catalytic pyrolysis, the majority of the feedstock metals are adsorbed on the catalyst surface. Thus, a spent catalyst may contain up to 23 wt.% Sb, up to 36 wt.% Ni, up to 26 wt.% Fe, and up to 28 wt.% Br.[160] Depending on the type of catalyst, up to 92% of the bromine in the polymer may pass to the solid residue.[161] Unfortunately, there are virtually no studies on ways to regenerate spent pyrolysis catalysts containing halogens and metals, and the traditional approach to regenerating cracking catalysts is unacceptable.
One of the uses of pyrolysis residue is gasification to produce synthesis gas and extract metals from the ash residue.[162] A significant part of research is devoted to the use of the solid product as an adsorbent for purifying water from drug impurities,[131] heavy metal salts,[163][164] organic pollutants,[165] and electrocatalysts.[143]
An important part of boosting the economic efficiency and environmental friendliness of the pyrolysis process is the use of all components of the gaseous product, the yield of which can reach 30% by weight (see Table 3). With an increase in pyrolysis temperature from 360°C to 440°C, the proportion of nitrogen passing into the gas phase (NH3 , HCN) increases.[118] During the pyrolysis of PAN, the content of NH3 and HCN in the gaseous product is higher than during the pyrolysis of ABS. The use of iron oxide-based catalysts helps to increase the proportion of nitrogen passing into the gas phase (Table 11).[117] The main hydrocarbon components of the gas phase include methane, ethane, ethylene, propane, propylene, butanes, butenes, and butadiene.[117][119][123][135] It is possible to use the gaseous product as a fuel or for the isolation of olefins.
Unfortunately, we were unable to find any studies devoted to NH3 and HCN recovery methods during the pyrolysis of acrylonitrile copolymers, although this area could be considered relevant and important for the development of nitrogen-containing plastic pyrolysis technologies. The release of HCl and HBr during pyrolysis inevitably leads to the formation of ammonium chlorides and bromides, which can form salt deposits on the surfaces of process equipment. The degree of nitrogen recovery will decrease due to its loss as inorganic salts.
7. Dehalogenation and denitrogenation of pyrolysis products
Even with catalytic pyrolysis, the Br content of the liquid product can reach several percent (see Table 8); it also contains phenols, Cl-, and N-organic compounds. The requirement for the absence of halogen and N atoms in petrochemical feedstocks dictates the search for product refining methods aimed at removing heteroatoms without reducing the content of olefins and aromatic hydrocarbons. This latter requirement precludes the use of hydrotreatment, since it will inevitably involve hydrogenation of styrene and α-methylstyrene, as well as partial dearomatization. Therefore, the methods for refining the pyrolysis product are reduced to extraction methods,[166] adsorption,[167] and hydrothermal treatment.[82][168] Hydrotreatment can be used if the pyrolysis product is intended to be used as fuel or for obtaining solvents (benzene, toluene, xylenes). Fig. 6 shows a possible scheme for dehalogenation and denitrogenation of the pyrolysis product.
The liquid pyrolysis product contains Br atoms in the form of organobromine compounds, SbBr3 , soluble in organic compounds,[123] and HBr, also soluble in organic products. Similarly, Cl is present in the form of organochlorine compounds and HCl. Distillation of the liquid pyrolysis product separates most of the dissolved HCl and HBr.[169] Thus, several fractions can be isolated from the liquid pyrolysis product of WEEE (Table 12). The Cl content in the fractions 1, 2, and 4 does not exceed 0.047 wt.%, with the initial content in the pyrolysis product of 0.4833 wt.%.[169] In the first three fractions, Br is present in trace amounts and is concentrated in the distillation residue and the 150 – 190°C fraction, which contains bromophenols. It can be summarized that distillation of the pyrolysis product allows for a significant reduction in the halogen content; however, the target fractions still contain Cl and Br atoms, making their use as petrochemical feedstock impossible without additional purification.
The feasibility of adsorption purification of pyrolysis products using activated carbon to remove nitriles, chlorobenzene, phenols, and other compounds has been studied.[167] However, this is limited to model systems; adsorption purification of actual raw materials, the pyrolysis product, has not been studied. Removal of HCl and HBr from liquid pyrolysis products is also possible using water extraction. The efficiency of HBr removal is close to 100%, but it significantly decreases in the presence of amines in the organic phase, likely due to the formation of bromide salts.
Organobromine- and -chlorine compounds cannot be separated by aqueous or alkaline extraction at room temperature; this requires hydrothermal treatment. Akimoto et al.[82][168] studied the hydrothermal treatment of the pyrolysis product of a mixture of WEEE and its kerosene fraction. The main nitrogen-containing compounds in this pyrolysis fraction were benzonitrile and caprolactam. Hydrothermal treatment using pure water at 200°C reduces the Cl content from 55 to 25 ppm, but denitrogenation requires a temperature above 300°C: at 425°C, the degree of denitrogenation was 89%. Changing the pH of the solution towards an acidic or alkaline reaction ensures more effective denitrogenation. Thus, hydrothermal treatment at a temperature of 350°C for 30 min in water decreases the N content in the kerosene fraction from 900 to 690 ppm, in a NaHSO4 solution — to 369 ppm, in solutions of NaHCO3, Na2CO3, Na3PO4 — to 143 – 166 ppm, in NaOH solutions —to 166 ppm, in KOH solutions — to 155 ppm. In a solution of 0.05 mol L–1 NaOH at a temperature of 300°C, exhaustive dechlorination is achieved; the N content in the product is 430 ppm and decreases to 40 ppm at 400°C (30 min). Therefore, the use of alkali solutions allows to reduce the temperature of hydrothermal treatment in comparison with pure water.
It should be noted that hydrothermal treatment in alkali solutions is preferable to that in solutions of acids and salts with a pH < 7, due to the fact that hydrolysis of nitrogen-containing compounds in alkaline solutions produces NH3 , which can be recovered (in a solution of 0.1 mol L–1 NaOH, a degree of NH3 recovery of 97% based on the initial N content in the pyrolysis fraction is achieved). Despite the high efficiency of dechlorination and denitrogenation of the kerosene fraction under hydrothermal conditions, the question of the possibility of denitrogenation of other, heavier fractions or the entire pyrolysis product without preliminary fractionation remains open. It was found[170] that nitrogen-containing heterocycles are unreactive under hydrothermal conditions at temperatures below 460°C. Light fractions from ABS pyrolysis contain N predominantly in the form of nitriles, while heavy fractions contain, inter alia, quinolines, pyridines, and pyrroles.[116][118] Hydrothermal treatment is likely justified for light fractions, for which hydrodenitrogenation is impractical due to styrene hydrogenation. For heavy pyrolysis fractions, however, hydrothermal treatment will be less effective, and the absence of styrene, toluene, xylenes, and ethylbenzene (valuable petrochemical compounds, the hydrogenation of which is undesirable) creates the preconditions for hydrotreatment.
Hydrodenitrogenation is an industrially proven process used to remove N atoms from gas oil fractions of petroleum,[171] coal distillates,[172] biomass pyrolysis products[173][174] and polymers.[175-177] Hydrodehalogenation is a well-studied process described in a number of review publications, including in relation to polychlorinated biphenyls,[178] and products from the recycling of chlorinated polymers,[179] and therefore will not be covered in this review.
In the hydrodenitrogenation of biomass and polymer pyrolysis products, sulfide catalysts on aluminum oxide (NiMoS/Al2O3),[173-176] or on activated carbon (NiMoS/C) are generally used.[177] Hydrotreatment of the WEEE pyrolysis product at a temperature of 350°C over the NiMoS/C catalyst[177] shows virtually exhaustive debromination, dechlorination and denitrogenation. Hydrotreatment of liquid pyrolysis products of tires[176] at a temperature of 362°C over a NiMo/Al2O3 catalyst provides a denitrogenation degree of 92% with an initial N content of 0.4 – 0.8 wt.%. The competition between hydrodechlorination and hydrodenitrogenation reactions in systems simulating the pyrolysis products of chlorine- and nitrogen-containing polymers was studied,[180] and it was shown that nitrogenous bases are undesirable components of the feedstock during hydrotreatment over tungsten and molybdenum disulfides, since they irreversibly deactivate the catalyst.
Unfortunately, there are no studies on the composition of the pyrolysis products of acrylonitrile copolymers after hydrotreatment, nor are there data on the degree of hydrogenation of aromatic hydrocarbons, including polycyclic ones. Based on these results, it would be possible to draw conclusions about the product’s potential uses: as a fuel, a raw material for extracting naphthalene and phenols, etc. Of current interest are studies of the parallel hydrodenitrogenation – hydrodebromination –hydrodeoxygenation reactions, and competition for active sites for various types of catalysts (platinum group metals, and transition metal sulfides, and phosphides) in the adsorption process. Unsupported molybdenum and tungsten disulfides, studied in the hydroconversion of petroleum residues, could presumably be used for the hydrotreatment of heavy pyrolysis fractions containing resins and asphaltenes.[181][182]
8. Conclusion
A major problem in the recycling of ABS, PAN, and NBR is the high N and Br content in the products and the need for their hydrotreatment, which, however, may result in the loss of valuable components such as styrene and α-methylstyrene. An analysis of the chemical methods for recycling acrylonitrile copolymers, schematically shown in Fig. 7, shows that pyrolysis is the most important technology; hydrothermal treatment is somewhat less studied and widespread. However, it is precisely in the WEEE recycling that hydrolysis and hydrothermal treatment are attractive in terms of preliminary preparation of raw materials[37] to remove some of the N and Br atoms and, accordingly, to reduce the content of organonitrogen and -bromine compounds in the pyrolysis product.
Hydrolysis of WEEE, including ABS waste, remains a novel solution, requiring the development of methods to increase the degree of debromination and denitrogenation. Research aimed at increasing the extraction of N and Br from plastic feedstock for subsequent use is also relevant. Research into catalysis under hydrothermal conditions, aimed primarily at intensifying the cleavage of carbon – heteroatom bonds, is of great importance for hydrothermal recycling.
For pyrolysis, as the most proven process for recycling plastic waste, the question remains as to creating catalysts that are resistant to deactivation, providing maximum removal of heteroatoms with minimal conversion of olefins and long-term operation without regeneration.
In terms of refining the products of pyrolysis and hydrothermal recycling of acrylonitrile copolymers, studies of both non-catalytic methods (hydrothermal processing, adsorption, extraction) and hydrotreatment are of great importance. It should be noted that hydrodebromination, in contrast to hydrodechlorination, has received little attention. The possibility of using traditional hydrotreating catalysts for processing feedstocks with high N and Br contents (up to 10 wt.%), resinous compounds, and phenols remains unresolved.
This review was written within the framework of the State assignment of the A.V.Topchiev Institute of Petrochemical Synthesis.
9. List of abbreviations and designations
ABS — acrylonitrile – butadiene – styrene copolymers;
ABS-Br — acrylonitrile – butadiene – styrene copolymer with brominated flame retardants;
EVA — ethylene – vinyl acetate;
HIPS — high-impact polystyrene;
MSW — municipal solid waste;
NBR — nitrile butadiene rubber;
PA — polyamides;
PAH — polycyclic aromatic hydrocarbons;
PAN — polyacrylonitrile;
PE — polyethylene;
PEG — polyethylene glycol;
PET — polyethylene terephthalate;
PMMA — polymethyl methacrylate;
PP — polypropylene;
PS — polystyrene;
PVC — polyvinyl chloride;
SAN — styrene – acrylonitrile;
TGA — thermogravimetric analysis;
THF — tetrahydrofuran;
TPD-NH3 — temperature-programmed desorption of ammonia;
WEEE — waste electrical and electronic equipment.
References